Published
A New Corrosion Protection System for Zinc and Zinc-Alloy Surfaces
This paper introduces a newly developed system for increased corrosion protection for zinc and zinc-alloy coated surfaces.
by Klaus-Peter Klos and Johannes Klaus
anjo Oberflächentechnik GmbH, Trebur/Astheim, Germany
anjo Oberflächentechnik GmbH, Trebur/Astheim, Germany
ABSTRACT
This paper introduces a newly developed system for increased corrosion protection for zinc and zinc-alloy coated surfaces. Here we present a set of cobalt-containing as well as cobalt-free passivates. The cobalt-free systems give improved white rust values, combined with cheaper chemicals. Cobalt-free systems are available as thin and thick layer passivates and as a black passivate. The black passivate works on alkaline or acid zinc and in combination with a proprietary nano-sealer, white rust values up to 120 hr can be easily obtained. The nano-sealer can also be applied to the blue and thick layer passivates. In combination with the cobalt-free thick layer passivates, red rust values over 720 hr can be obtained, which meets most car manufacturer requirements. The combination of plain zinc, a thick layer passivate and the nano-sealer is an easy-to-use system and in comparison to zinc/nickel, money can be saved. Beside the regular passivates, we will discuss a colorable passivate in which dyes are dissolved. This passivate produces a large variety of different colored finishes, for example on red or green screws.
Keywords: cobalt-free passivates, cobalt-containing passivates, nano-sealers, zinc and zinc alloy post-finishes
Introduction
In recent years, we have seen major changes in our industry. We have had to replace hexavalent chromium (Cr+6) in all chromates and a significant portion of zinc plating has been shifted to alloy plating, primarily to zinc-nickel. The first move (from Cr+6) was initiated by the ELV (End of Life Vehicle) regulations from the European Union. The second one (to Zn-Ni) addressed the need for higher corrosion resistance. The first step was achieved by replacing Cr+6 with cobalt. Since the recent actions to classify cobalt as toxic and carcinogenic, many platers have been seeking cobalt-free passivates. In the case of zinc-nickel-plating, there are concerns about the high cost and the uncertain future of nickel, particularly from a regulatory standpoint.
Furthermore, the difference in corrosion resistance between zinc and zinc-nickel is quite significant. Under normal plating conditions, the corrosion protection of zinc is below 500 hr to red rust (Fig. 1). By contrast that of zinc-nickel is far higher, exceeding 720 hr. A German car manufacturer put this into the following words: “We put 720 hr into our specifications because we do not want to test any longer.”
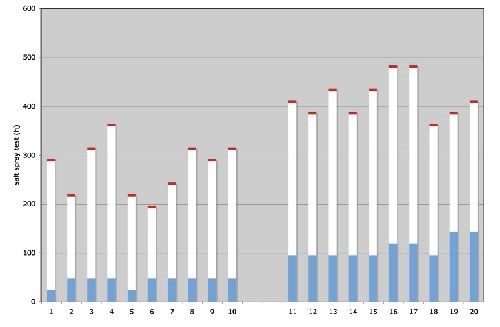
Figure 1 - Corrosion protection of zinc plated parts via neutral salt spray test:
(L) (1-10): washers, 3.5 μm zinc, cobalt-free thin film passivate;
(R) (11-20): screws, 8.3 μm zinc, cobalt-free thick film passivate;
Blue = no corrosion; white = white corrosion; red = red rust.
(L) (1-10): washers, 3.5 μm zinc, cobalt-free thin film passivate;
(R) (11-20): screws, 8.3 μm zinc, cobalt-free thick film passivate;
Blue = no corrosion; white = white corrosion; red = red rust.
Figure 1 also shows that even with a thick film passivate and higher zinc thickness (right sample) we cannot achieve more than 500 hr to red rust. Zn-Ni alloys (12-15% Ni) achieve more than 720 hr. Another advantage of a Zn-Ni alloy is the stability of the corrosion protection when the parts are exposed to higher temperatures up to 200°C.
Today we can offer our customers a new system, based on zinc, a passivate and a nano-coating sealer (ZPN). This system uses all common types of zinc plating (acid, alkaline, cyanide) as well as zinc alloy processes. The passivates are cobalt-free and the color can be silver/blue or black. The last step of the process is a ceramic coating based on silicon dioxide.
The key to better corrosion protection is the nano-coating in combination with a compatible passivate. Not every passivate can be used. Practical tests must be done to qualify the passivate.
The nano-coating is applied in a dip-spin unit. The dipping time is 10 to 20 sec. The spinning time is less than 60 sec at high speed. A tilting during spinning is recommended. The spinning is followed by a backing process equal to or above 120°C (object temperature).
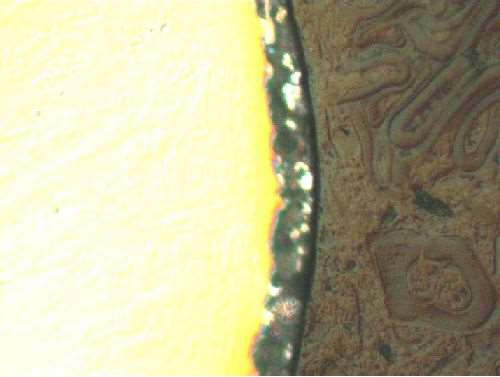
The deposit thickness ranges from 0.5 to 1.0 μm (500 to 1000 nm) (Fig. 2). It is very uniform as can be shown by adding a dye to the nano-coating (Fig. 3). The parts show a uniform red coating on a colorless surface. Since the thickness is so thin, the coating can be applied on very small parts. One of our customers has done coatings on M0.6 screws with no problems.
The complete system exhibits very high corrosion resistance. Figure 4 shows the results for ten washers, plated with 3.5 μm zinc, a cobalt-free thin film passivate and the nano-coating. The parts were made in a production line with the barrel load capacity of 50 kg. After the backing process all washers were easily separated, with no sticking together.
Figure 4 also shows the results for ten smaller screws (right side of figure). In this case, the passivate was a cobalt-free thick film passivate. This combination achieves more than 720 hr to red rust. Figure 5 shows the final results for these screws after 1800 hr of neutral salt spray testing. All tests were carried out under the DIN EN ISO 9227 neutral salt spray test.
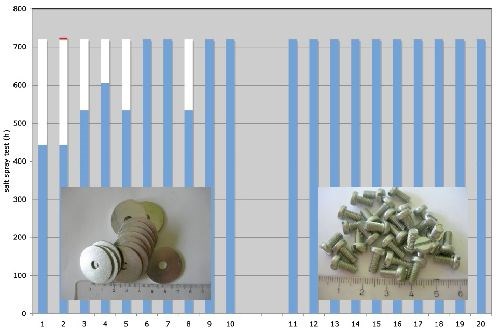
Figure 4 - Corrosion protection of zinc plated parts with nano-coat via neutral salt spray test:
(L) (1-10): washers, 3.5 μm zinc, cobalt-free thin film passivate + nano-coat;
(R) (11-20): screws, 8.3 μm zinc, cobalt-free thick film passivate + nano-coat;
Blue = no corrosion; white = white corrosion; red = red rust.
(L) (1-10): washers, 3.5 μm zinc, cobalt-free thin film passivate + nano-coat;
(R) (11-20): screws, 8.3 μm zinc, cobalt-free thick film passivate + nano-coat;
Blue = no corrosion; white = white corrosion; red = red rust.
The minimum temperature of the backing process is recommended to be 120°C. Nonetheless, higher temperatures up to 200°C and longer backing times can be used. Thus the process for hydrogen release can be used as backing process as well. In this case the parts can be done in a single process in a plating line.
We have also had the opportunity to study the addition of different dyes to the passivate. Figure 6 shows some of the colors obtained: blue, green, yellow and red. The process is a one-step process. The parts can be coated with the nano-coating as well. The color is temperature stable and does not leach out.
The ZPN-system is a good method to improve the corrosion protection on a zinc or zinc-alloy surface. With good temperature stability and the excellent corrosion resistance, it is in some cases a good alternative to zinc-nickel. We know that not all zinc-nickel applications can be replaced (like those in contact with aluminum), but in many cases this system is less costly and an applicable replacement.
Reference
1. Dr. Hesse, Daimler-Benz-AG, Germany, Verband der Automobilindustrie (VDA) Meeting (German Association of the Automotive Industry), November 11, 2009.