Getting Better All the Time
Trivalent black passivates currently in use reflect the great strides made in appearance, corrosion protection, and solution life. All these are improved and the processes are a little more like the old two-part hex black. However, they still employ multiple additives and controllers to maintain the solution.
#pollutioncontrol
Innovation and trend-setting technology have always driven suppliers of zinc plating processes. To “future-proof” the survival of the zinc plating industry, we must be able to provide customers with the products they need now as well as innovations they will need many years down the road. This article provides a look at some technology advancements either just released to the market or currently under development at our company. Consider these forward-thinking strategies an effort to sail smoothly through any “future shock.”
Next-Generation Tri Black Chromate
Featured Content
To find out where we’re heading with trivalent black chromates, we first have to see where we’ve been and where we are. Prior to the European Union’s End-of-Life Vehicle (ELV) directive and the restriction of hexavalent chromium on finished zinc-plated parts, the two-part silver-Cr6+ was the staple chromate for producing a black finish. Although expensive, somewhat labor-intensive and at times inconsistent in color, the two-part hex black had a successful run dating back to the late 1940s. When the two-part hex black chromate solution was tweaked just right and parts were processed by an experienced “dipper,” the finish was jet black with a high-intensity gloss.
With the demise of hexavalent chromates, first-generation trivalent black passivates came to market in early 2000. These processes had multiple problems, including poor corrosion protection, unsatisfactory appearance and color, and short solution life.
Trivalent black passivates currently in use reflect the great strides made in appearance, corrosion protection, and solution life. All these are improved and the processes are a little more like the old two-part hex black. However, they still employ multiple additives and controllers to maintain the solution. This can become labor-intensive due to the constant tweaking required to produce an acceptable and consistent black finish. Line operators are required to perform numerous analyses each shift to determine chrome content and operating levels in the tanks. After laboratory analysis, a somewhat unscientific approach of guessing and juggling of additives is needed to “bring the solution back.” Table 1 lists the attributes of second-generation trivalent black passivates.
|
Table 1: Second-Generation Trivalent Black Chromates
|
|
|
Attribute
|
Comments |
| Appearance | Improved gloss-black and scratchability, still some inconsistency |
| Film thickness | 100–200 nm |
| Performance | 96+ hr to first white rust |
| Chromate additives | Still require multi-component additives, polymer and chrome-based sealers |
| Solution life, Maximum zinc content, ppm |
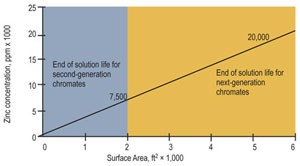
Improved; may last a couple of days to many weeks 5,000–8,000 |
| Notes | Improved polymer and chrome-based sealers, Better process stability, Continue to be complicated and laborious to run consistently |
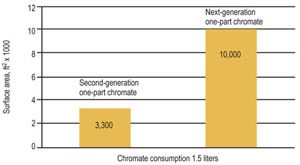
The key to developing the next generation of trivalent black passivates will be to create a true one-component chromate that is easy to use and maintain, such as Columbia Chemical Corp. just-introduced TriVecta UniBlack. This new trivalent black passivate offers what all future passivates will be required to provide—a consistent gloss black appearance, high corrosion protection and tolerance to zinc build-up for economical operation. Table 2 compiles requirements and a “wish list” for the next-generation one-part black passivate.
One of the biggest complaints from users of today’s trivalent black passivates is the inability to run the process on a daily basis without constantly babysitting the solution or requiring the supplier to camp out at the shop to maintain the solutions. A true one-component chromate has to be stable and contain all trivalent chromium salts, acid salts, metal salts and other proprietary chemicals as one “make-up/starter” product and as the replenisher.
|
Table 2: Next-Generation One-Part Black Trivalent Chromate
|
|
| Attribute | Comments |
| Appearance | Uniform gloss-black, scratch-resistant |
| Chromate film thickness, nm | 400+ |
| Salt-spray performance | 144+ hr to first white rust |
| Chromate additives | True one-part chromate and one-part sealer |
| Solution life | Excellent life, cost-effective chemical replenishment |
| Maximum zinc content, ppm | 20,000+ |
| Notes | Wide operating window (pH, temp, immersion time), Very easy to use, no juggling components, True reactive sealer reacts with chromate film |
Zinc build-up has always been a chromate killer, regardless of whether it was in hexavalent or trivalent chromates. Since trivalent chromates are much more sensitive, new technologies must slow down the build-up of zinc and increase tolerances of the dissolved zinc metal. As zinc builds, higher levels of chromate additives are required to compensate for this contaminant growth. Zinc lowers corrosion protection and causes the finish to have a mottled gray appearance.
Another critical component of future one-part trivalent black processes is chromate “mileage.” The present two-part trivalent black passivates require high replenisher additions to maintain salt-spray performance and appearance. This is due to the low oxidation state (inability to promote chromate film growth), depletion and precipitation of key process components, characteristics of the zinc deposit (leveling, brightness, thickness) and hypersensitivity to solution temperature and pH.
Cobalt-Free Chromates
With the painful yet successful switch from hex to trivalent chromates, the need for even more environmentally friendly passivates is on the horizon. The EU’s Restriction of Hazardous Substances (RoHS) directive requires periodic review of new scientific evidence, with the intent of adding potentially hazardous substances to the prohibited list.
Cobalt is being considered as a “high-priority substance/material” for addition to the list of RoHS prohibited substances. Selection of these high-priority hazardous substance candidates is based on criteria that addresses both environmental and human health impacts. Environmental impact includes the formation of hazardous substances during collection and treatment of electrical and electronic equipment or during recycling or incineration. The impact on human health encompasses whether or not the substance has carcinogenic, mutagenic or toxic properties.
If and when cobalt is moved from the RoHS watch list to the active prohibited substance list, other expensive, exotic substances will be needed to meet industry specifications. This is evident from our experience with the elimination of hexavalent chromium, when more expensive substances such as trivalent chromium, cobalt and chelating acids were required to meet automotive and general zinc plating specifications. Avenues now being explored to replace cobalt include:
- Formulating trivalent chromates with integrated silica compounds
- Adding specially formulated organic compounds
- Adding organo-metallic compounds
- Adding expensive transition metals.
Looking back at the initial changeover from hexavalent to trivalent chromates, elimination of cobalt will surely increase product costs and force chemical suppliers and plating operators to invest the necessary time to master the non-cobalt learning curve.
No Nitric or Nitrates
Nitric acid and its related acid salts are also on the potential RoHS hit list due to U.S. Environmental Protection Agency (EPA) National Primary Drinking Water Regulations, extremely hazardous substance listing requirements, and other pending regulations.
Nitric acid and its related acid salts has been workhorses for chromate technologies for many, many years. Nitric acid activates the zinc deposit surface, promotes chromate film growth and provides clarity and color. For this reason, future R&D efforts must center on formulations to replace this key component.
Total elimination of nitric acid from trivalent chromates will increase use of other acids and their salts, such as sulfuric, hydrochloric and hydrofluoric. Chelating and non-chelating acids will also need to be incorporated into new formulations. As with most substances that become regulated, non-nitric acid trivalent chromates will be initially more expensive, more difficult to use, and have appearance and performance problems.
Improved Zn-Ni Alloy Processes
As zinc-nickel alloy plating continues to grow, improvements in appearance, brightness, and chromate receptivity are required to satisfy this higher-end market. Table 3 highlights deficiencies and areas for improvement needed for today’s alkaline zinc-nickel alloy process.
|
Table 3: Characteristics of Current Alkaline Zinc-Nickel Alloy Plating Processes
|
|
| Appearance | Matte to moderate bright |
| Chromate receptivity | Can produce dark, discolored, mottled finish |
| Deposit distribution | Acceptable; better than acid zinc-nickel |
| Nickel distribution | Excellent over a range of current densities |
| Cathode efficiency | Low; slow plating speed |
| Operating range | Narrow zinc, caustic, temperature and proprietary additive range |
| Notes | Sensitive to carbonate build-up, Inability to treat contaminants |
|
Table 4: Next-Generation Alkaline Zinc-Nickel Alloy Plating
|
|
| Appearance | Full bright |
| Chromate receptivity | Provides true chromate color and appearance, does not darken or mottle appearance |
| Deposit distribution | Excellent, comparable to present advanced alkaline zinc processes |
| Nickel Distribution | Maintain over a range of current densities |
| Cathode Efficiency | High, comparable to present advanced alkaline zinc processes |
| Operating Range | Must have a wide zinc, caustic soda, temperature and proprietary additive range |
Low cathode efficiency (slow plating speed) is a primary target area for future product development. Probably the biggest complaint today from platers is low bath efficiency, which equates to slow plating and low output. Platers want the high efficiency, quick plating and high output they presently get from their alkaline zinc (non-alloy) plating lines, which have much higher cathode efficiency and deliver more than 50% more output. In today’s ultra competitive zinc market, this is a tremendous benefit and profit maker.
Future zinc-nickel alloy chemistries will have to deliver at least the same efficiency and output presently found in today’s advanced alkaline zinc processes. Table 4 provides additional requirements for next-generation alkaline zinc-nickel alloy processes.
Although acid zinc-nickel alloy plating is a much smaller market than alkaline zinc-nickel, it has one nice attribute: its ability to plate heat-treated and hardened steel. The process’s high cathode efficiency (90%+) provides two things: immediate deposit initiation and fast plating speed. In the alkaline bath, parts gas heavily and polarize before initiating the first phase of zinc deposition. This can take up to 10 min before even the first layer of zinc-nickel starts to deposit on the part. Acid baths, in contrast, start plating and laying down zinc-nickel as soon as the part is hit by current.
But unlike alkaline alloy processes, acid zinc-nickel bath has poor deposit distribution that makes it unsuitable for most rack applications. Because a “true throwing power/covering power” process for non-alloy acid zinc has yet to be developed, it is going to be a real struggle to develop one for acid zinc-nickel—development would be much easier if there was already a platform to work from. Regardless, throwing power, even if it is on a micro or limited basis, is another area that must be investigated and improved.
Table 5 is a summary of present-day acid zinc-nickel alloy plating, while Table 6 outlines areas for improvement in a next-generation chemistry.
|
Table 5: Characteristics of Current Acid Zinc-Nickel Alloy Plating Processes
|
|
| Appearance | Matte to semi-bright |
| Chromate receptivity | Can produce dark-dull-satin appearance |
| Plate distribution | Poor, comparable with present acid chloride zinc processes |
| Nickel distribution | Fair, deposits more nickel at higher current densities |
| Notes | Anodes polarize, Requires elevated bath temperature due to high chloride content Solution easily precipitates, difficult to maintain filtration |
|
Table 6: Wish List for Next-Generation Acid Zinc-Nickel Alloy Plating
|
|
| Appearance | Full bright, comparable to acid chloride zinc |
| Chromate receptivity | Provides true chromate color and appearance, does not darken or mottle appearance |
| Plate distribution | Provides some covering and throwing power |
| Nickel distribution | Maintains over range of current densities |
| Notes | No polarization of anodes, Economical to operate, does not require elevated temperatures or bath chemistry |
Summing Up
Upcoming changes and environmental/human health mandates relating to zinc plating can be viewed as either an impending disaster for our industry or a fantastic opportunity for suppliers. Without doubt, the change from hexavalent passivates to trivalent passivates was difficult, labor-intensive and expensive. However, the zinc plating industry as a whole became much better.
The same will hold true with the next round of prohibited substances. New processes will create a win-win situation for the environment and for human health, and will also raise the bar on performance requirements for finished zinc-plated components.
RELATED CONTENT
-
Copper Plating on Aluminum and Aluminum Alloys
How can I plate copper on aluminum?
-
Aluminum Anodizing
Types of anodizing, processes, equipment selection and tank construction.
-
Masking for Surface Finishing
Masking is employed in most any metal finishing operation where only a specifically defined area of the surface of a part must be exposed to a process. Conversely, masking may be employed on a surface where treatment is either not required or must be avoided. This article covers the many aspects of masking for metal finishing, including applications, methods and the various types of masking employed.