Nanotechnology in Finishing
Very small particles are having A Very large impact on coating materials and processes
You may think nanotechnology is just a buzzword. Or you may think it’s a laboratory phenomenon, with practical applications years if not decades down the road. You may even subscribe to the idea that out-of-control nanotechnology will result in humanity and the rest of the Earth eventually being enveloped in a “gray goo” of self-replicating nanobots, a scenario proposed by several science (or science fiction) writers.
Nanobots aside, the reality is: Nanotechnology is here, and finishers can reap the benefits of some of the first practical applications of nanomaterials science. Use of nanomaterials in coatings can not only enhance such “conventional” properties as hardness, scratch-resistance and corrosion resistance, it can also result in coating materials with novel insulating, radiation transmitting, and other properties.
Featured Content
Here’s a look at the current state of nanotechnology as it relates to coatings, and how it is changing the development of both organic coating materials and plating chemistries.
Nanotech: What is it?
The prefix “nano” is used in science to mean one billionth—a nanometer, for example, is one-billionth of a meter. Anything on that scale is so small as to be difficult to even imagine, but to attempt to put it in perspective, a human hair ranges from 60–120 μm in diameter—thousands of times larger than many nanoparticles used in coatings today. At such scales, the rules of physics and chemistry that govern the macro world no longer apply
According to Jamil Baghdachi, professor and director of the Coatings Research Institute at Eastern Michigan University (Ypsilanti, MI), recent work has had to overcome several obstacles to allow practical use of nanoparticles, including issues involving production, handling and measurement of the materials. “In the past, we did not even have methods to produce nanoparticles,” he says. “We’ve been able to do that using a variety of techniques for the past several years.
“The problem is, these particles tend to stick to themselves,” Baghdachi continues. “So, we’ve found ways to mechanically pulverize them and then separate them in such a way that they cannot stick together anymore. And, these particles are so small there’s really no way to detect them, to see them. You can’t use an optical microscope; the wavelength of visible light is 400 nm, and we’re talking about particles much smaller than that. So measurement of nanomaterials does not fall into our ‘normal’ range.”
According to Baghdachi, the same is true of nanomaterial properties. “In conventional coatings, for example, we look for things like hardness and color,” he says. “We can’t use these methods to describe the properties of nanomaterials. Instead, we have to look at the combined effect of nanomaterials on coating performance as opposed to their individual properties.”
Baghdachi says one of the first inroads of nanomaterials in the finishing industry has been providing improved scratch resistance in organic coatings. “A big issue today, particularly in the automotive industry, is scratch resistance,” he says. “Scratch resistance of paint can be improved by incorporating nanoparticles of aluminum oxide or silica. Those materials are hard, and they are so small that light passes through easily. Effectively, they’re transparent.”
Better scratch resistance is only one of the property improvements seen in organic coatings as a result of nanotechnology additives. Researchers at the University of Warwick (England) Department of Chemistry recently found a way to replace the soap typically used to stabilize latex emulsion paints with a nanotech clay material they say results in a paint that’s not only much more durable but also fire-resistant.
The scientists say they developed a simple way to individually coat the polymer particles used in paint with a series of nanosized clay discs, effectively creating an “armored” layer on individual polymer latex particles in the paint. The 1-nm thick, 25-nm diameter clay discs can be applied using current industrial paint manufacturing equipment, according to the researchers.
Nanometal Oxides
Fire-resistant paint formulated in a research lab is one thing, but closer to commercialization are single- and multi-metal oxide nanoparticles manufactured by NanoProducts Corp. (Longmont, CO). According to VP Rich Zvosec, one of the main applications for the particles is as a paint additive.
“In coatings, we’re focused on three areas: infrared absorption, UV blocking, and matching or altering the refractive index of materials,” Zvosec says. “Our nanoparticles result in transparent materials that absorb infrared light. That can be useful, for example, in coatings that are slow to dry or cure in infrared ovens due to their low absorption of near to medium IR wavelengths.”
“We’re also looking at UV blockers,” he continues. “Nanometal oxide UV blockers provide reduced yellowing, greater clarity, lower photocatalytic activity and easier dispensability than zinc oxide, and have a longer life than the organic UV blockers in use today.”
A third area under investigation is modifying the refractive index of a material to obtain, for example, higher gloss from a coating. According to Zvosec, his company is working on commercial applications in all three areas with about 15 different partners, including formulators and end users.
NanoProducts manufactures the particles using a high-temperature gas-phase process. Zvosec explains the company uses plasma torches to combust organo-metallic precursor materials in reactor vessels. “As the metal ions are created in an oxygen atmosphere, they condense out into metal oxides,” he says. “Particle size ranges from 10–200 nm, and particle composition, crystal structure, size and size distribution can all be controlled by changing process parameters.”
Zvosec says his company’s nanotechnology metal oxides also allow loading at levels far lower than those used with conventional additives. “Nanomaterials cost a lot more than conventional fillers, but our materials are used at very low loadings. We are looking at addition levels of 100 ppm up to about 1–3%, which would be the maximum for most of these materials,” he says.
Insulate & Encapsulate
Industrial Nanotech Inc. (Naples, FL) is another relatively small company with big ideas based on its proprietary nanomaterials technology. The company also develops its own coating formulations for several applications, including insulation against heat and cold and lead encapsulation.
According to VP Francesca Crolley, the company was born a few years ago out of CEO Stuart Burchill’s desire to develop practical applications for nanotechnology. “He was familiar with the coatings industry, so developing technology in that area was a natural fit,” Crolley says.
The company’s main products are its patented Nansulate line of coating materials, which are based on a proprietary particle called Hydro-NM-Oxide. The particles themselves are not true nanomaterials; they are micron-size. But their performance characteristics are the result of their nanoscale internal structure and surface chemistry.
Industrial Nanotech closely guards details of the particles’ production process and composition, but the company explains that the particles get their insulating properties from an internal structure of tunnels that heat must navigate in order to pass through the coating. Fully cured, Nansulate coatings consist of more than 70% by volume of the nanomaterial. Conduction of heat is hindered by the tiny size of the connections between the particles making up the conduction path, and the solids that are present consist of very small particles linked in a 3-D network with many “dead-ends.” Thermal transfer through the solid portion thus occurs through a very complicated maze and is not very effective.
The products are based on a water-based latex carrier system, and are low-VOC and non-toxic, according to Crolley. They are also very easy to apply, she adds. “That’s part of the beauty of the product. Even homeowners can apply it themselves and gain the benefits.” The company says Nansulate materials also provide excellent protection against rust and corrosion.
One application where stability of temperature and other process parameters is important is reactive dyeing of cotton. According to one user of Nansulate materials, reaction temperature during the process oscillates below and above the reaction set temperature, decreasing reproducibility in the color of dyed items. Insulation of the machine and piping can alleviate the problem, but it’s difficult to accomplish on the large, cylindrical dyeing machine itself.
For a dyeing process that takes place at 60°C, application of Nansulate material to the machine and process piping resulted in a reduction in cycling of a steam-based heat-exchanger from 24 heat-on cycles to two heat-on cycles during a one-hour processing time. Temperature variation also decreased, and fabric color from one batch to another was more consistent.
Another product from Industrial Nanotech is aimed at abating problems caused by lead-based paints. Nanotech LDX has already been used to provide cost-effective lead abatement in building renovation projects. The transparent product can be applied to a wide variety of surfaces, including wood and brick.
More recently, the U.S. Navy began trials of the patented product, which is being applied at a Navy facility in the Pacific.
Nano + UV = Novel Coatings
Ecology Coatings Inc. (Akron, OH) has made a name for itself by combining nanotechnology with UV curing to provide organic coatings with novel properties. According to the company, its “liquid nanotechnology” results in 100% solids, UV-curable coatings that use 80% less energy, require 80% less floor space, reduce emissions, and provide performance better than traditional carrier-based coatings.
Ecology says nanoparticles can be used in 100% solids-UV curable coatings to improve rheological properties, resulting in improved leveling and edge coverage when finishing metal parts. Nanoparticle additions are also said to improve hiding power of coatings. In one test, incorporation of nanoparticles resulted in a coating material with hiding power at thickness of 1.2 mils equivalent to that of a similar black, pigmented coating 1.5 mils thick.
The company says corrosion resistance of metal parts can also be improved by using both nanosize and micron-size particles in the same coating formulation. The intermingling of particles is said to effectively fill spaces, blocking entry by water and air and filling tiny flaws.
According to Ecology Coatings, nanoparticles can be used in both high-gloss and flat coatings. The company has developed techniques to disperse nanoparticles in monomer components to produce liquid components for the matting of 100% solids UV-curable coatings.
A Unique Process
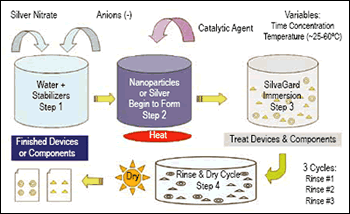
One nanotechnology-driven process that pretty much defies classification as an organic coating or plating process is a nanometal deposition process developed by AcryMed Inc. (Portland, OR) and first introduced to Products Finishing readers in last month’s issue (see the article “Nanotech Silver Stops Microbes” on page 18).
Company CEO Jack McMaken says his company worked first on medical applications for its nanotechnology coating process because that’s the application area with which managers were most familiar. “Our initial focus, because we’re medical device guys, has been on developing the silver for antimicrobial purposes,” he says. “But the coatings that result from this process are also conductive, they can be linked with organics, and there are optical applications as well. For example, the nanoparticles tend to settle into microscopic valleys on the surface being treated first, so it’s potentially applicable for developing super-smooth and reflective surfaces for optics.”
And, the process is not limited to a single nanometal. According to McMaken, the AcryMed process offers a unique method of generating and attaching metallic nanoparticles onto surfaces. “The process is based strictly on a size phenomenon; it’s not tied to a particular material,” he says. “So the particles don’t have to be silver. We can also produce nanoparticles of gold, platinum and copper, and the process works well with all of them.
McMaken says AcryMed is open to working with partners interested in the process for applications outside medical device manufacturing.
“For possible electronics applications, for example, we can load the substrate up until it becomes conductive and carries a current,” he explains. “We’ve even done this with elastic substrates like silicone. When you stretch this stuff, the particles don’t fall off. They basically spread apart, and after you stretch it two or three times you lose conductivity because the particles don’t touch each other any more. But, when you let it come back, the conductivity comes back. So that tells us that these particles are individually attached to the surface.”
Inorganic Coatings
Overall, use of nanotechnology in inorganic coatings technologies such as plating seems to be less advanced than in organic coatings. That doesn’t mean that nothing is happening, or that suppliers of plating chemistry and processes are not interested in the technology. In fact, all of the plating technology suppliers initially contacted for this article said they were working with nanotechnology. It’s just that, at this stage, very few are willing to talk about their activities on the record.
But that doesn’t mean there’s nothing to report. Researchers at the University of Arkansas (Fayetteville), for example, recently announced development of a simple, inexpensive way to create a “nanowire” coating on the surface of biocompatible titanium that can be used to create more effective surfaces for hip replacement, dental reconstruction and vascular stenting. The material can be easily sterilized using ultraviolet light and water or using ethanol.
The researchers used an alkali and heat to create titanium oxide-based ceramic nanowires that coat the surfaces of titanium alloy medical devices. Nanowire length and height, as well as pore openings and pore volumes within the nanowire scaffolds, can be controlled by varying the time, temperature and alkali concentration in the reaction, the scientists say.
Passivation with Nanoparticles
Laboratory production of ceramic nanowires using a process akin to plating is certainly interesting and someday may be important in medical device applications. But one technology area of more practical value for many platers and where use of nanoparticles has already had a significant impact is in passivates or conversion coatings used after zinc plating processes. This is especially true for trivalent passivates, which allow platers to enhance the corrosion resistance of zinc-coated parts without the environmental and worker safety issues surrounding use of hexavalent chromium.
Several suppliers have already developed such materials, including Pavco, Inc.(Charlotte, NC). Michael Marzano, VP and technical service director, says his company’s trivalent chromate passivate chemistries include nanoparticles ranging in size from 2–22 nm and in surface area from 60 to almost 500 m2/g. “We have found that adding these particles can increase salt-spray corrosion protection of finished parts by 50–100 hr. We think the nanoparticles build up within the coating and produce a physical barrier between the coating and the environment to extend corrosion protection,” he says.
According to Marzano, Pavco is working with multiple types of nanoparticles, including fumed silica and several types of clays, in an attempt to incorporate nanotechnology in some newer chemistries under development at the company’s Compliant Chemistry Division in Independence, OH.
Other suppliers of trivalent chromium passivates also use nanoparticles in their formulations to achieve similar results.
Nanocrystalline Metals
One company that has been involved in combining nanotechnology with electroplating since the mid-1980s is Integran Technologies Inc. (Toronto, ON). According to CEO Gino Palumbo, his company has worked on taking the next logical step in materials science by focusing not just on what elements are in a material but how they are arranged on a nano scale. The result is more than 100 patents in the areas of nanomaterials and processing.
“In the past, most significant changes in material performance have come about as a result of changes in alloy chemistry,” Palumbo explains.
“But equally important is how the atoms of various materials actually come together inside the material itself. So essentially what we do is modify basically conventional materials’ production processes to try to optimize the internal structure of the material. Rather than wholesale chemistry changes, we focus on modifying how the alloy is made.
“It’s kind of like building a brick house,” he says. “The material the bricks are made of is very important, but equally important or even more important is how you stack those bricks together.”
Integran develops nanotechnology metals using two processes: grain boundary engineering and electroplating (or, for production of larger parts or bulk materials, electroforming).
According to Palumbo, grain boundary engineering involves taking conventional metalworking processes such as rolling, forging and heat treating and modifying the amount of work done and the processing temperatures to obtain the desired grain structure in the material.
“Grain boundaries—the interfaces between crystals in a metal—have traditionally been weak points, subject to preferential corrosion and cracking,” he says. “We modify the metalworking processes to actually obtain a better fit between crystals, which leaves less space at the interfaces and makes the grain boundaries a lot more resistant to corrosion and cracking.”
Palumbo says grain boundary engineering works because all materials want to find their lowest-energy state.
“In most conventional metalworking processes, the focus is on getting the product beaten into shape as quickly as possible, getting it heat treated and getting it out the door,” he explains. “We modify the processing to allow more time for the material to achieve its lowest energy configuration at each step in the process and fill in the holes between the crystals.”
Probably of more interest to platers are the company’s nanotech plating processes and chemistries, which have been shown to perform with multiple metals and alloys. “It’s been known for many years that making grains smaller in a material greatly enhances mechanical properties, so we started thinking about how to refine grain size to the extreme,” Palumbo says. “We discovered we could do that using basically a conventional electroplating process. By modifying plating bath chemistry and the current wave forms we apply, we can promote nucleation and hinder growth of crystals to produce nanocrystalline materials.
“The beauty of this approach is, electroplating is a process that’s well known and understood and used around the world, and our technology is almost a direct drop-in replacement for companies interested in producing nanocrystalline materials.”
According to Palumbo, Integran Technologies, Inc. has used the electroplating/electroforming process to produce a range of alloys and pure metals with controlled grain sizes down to about 10 nm. “When you do that, you get very significant improvement in hardness and strength—typically about a five- to seven-fold increase,” he says. “That’s huge, and typically you wouldn’t be able to achieve that just by changing alloy chemistry.”
One of the first materials developed using the technology was Nanoplate cobalt alloy, a potential replacement for hard chromium in wear and corrosion applications. Developed under a contract with the U.S. Department of Defense, the process is commercialized in hydraulic component applications and is now nearing final qualification and approval for aerospace and defense applications.
“The process actually will be an integral part of the Joint Strike Fighter program, primarily for coating landing gear components,” Palumbo says. “The alternative was high-velocity oxyfuel thermal spray, which is a line-of-sight process. Our process can coat internal surfaces you can’t get to using thermal spray.”
According to Palumbo, the cobalt metal used in the process is substantially more expensive than chromium, but process efficiencies more than make up the difference in initial cost. “In chrome, the base metal is usually a relatively small component of the cost of the coating,” he says. “Cobalt is more expensive, but the Nanoplate process is very efficient and there are fairly substantial operating cost savings in terms of efficiency and improved throughput that can come from adopting the technology. Hex chrome runs at about 20% electrical efficiency. Our process runs close to 90–95%. So there’s a potentially significant increase in throughput.”
Not to mention elimination of hex chrome and the environmental and worker safety issues that accompany it. “We haven’t played up the environmental and worker safety angles too much because many of the companies we are dealing with have already bit the bullet and made substantial investments to deal with those issues, but you do eliminate hex chrome,” Palumbo says.
Palumbo says Integran’s cobalt Nanoplate and other nanotechnology plating processes may be relatively easy to implement for finishers using conventional electroplating chemistries. “Our philosophy has always been to make it easy for platers to adopt a chemistry change, and our processes are essentially drop-in replacements for conventional electroplating processes,” he says. “The worst case would be a small capital investment for a certain type of rectifier that we would specify. Sometimes, you can use existing rectifiers with some add-ons, and sometimes it’s a straight drop-in replacement. But we provide support through the adoption cycle, from identifying which of our materials will perform the function customers want to making some parts for testing and development to quantifying the cost impact for adoption.”
To speed penetration of the technology in key industries, Integran Technologies, Inc. has set up three companies to commercialize its materials in the sporting goods, aerospace and automotive industries. An early success in a sporting goods application is use of a nickel nanomaterial coating on Epic brand carbon composite golf club shafts made by Grafalloy (Memphis, TN).
According to Power-Metal Technologies (Carlsbad, CA), Integran’s licensee in the sporting goods area, the nanometal coating acts as a hard but ductile protective layer on the carbon composite material to limit potential impact damage and crack propagation, improve the shaft’s life and increase the power delivered with each swing. The lightweight shafts have more than twice the yield strength of steel or carbon composites and maintain the vibration-damping qualities of composite shafts, according to Grafalloy.
RELATED CONTENT
-
Masking for Surface Finishing
Masking is employed in most any metal finishing operation where only a specifically defined area of the surface of a part must be exposed to a process. Conversely, masking may be employed on a surface where treatment is either not required or must be avoided. This article covers the many aspects of masking for metal finishing, including applications, methods and the various types of masking employed.
-
Stripping of Plated Finishes
The processes, chemicals and equipment, plus control and troubleshooting.
-
Smut and Desmutting
Question: I am new to this industry and have heard about smut and desmutting operations.