Research and Decorative Coatings - The 19th William Blum Lecture
This article is a re-publication of the 19th William Blum Lecture, presented at the 65th AES Annual Convention in Washington, DC, on June 26, 1978.
#research
by
Burton B. Knapp
Featured Content
Recipient of the 1977 William Blum AES Scientific Achievement Award
Editor's Note: Originally published as Plating & Surface Finishing, 65 (8), 24-30 (1978), this article is a re-publication of the 19th William Blum Lecture, presented at the 65th AES Annual Convention in Washington, DC, on June 26, 1978. A printable PDF version is available by clicking HERE.
In selecting a subject for this lecture, I found it was difficult to deal with any phase of electroplating without encountering some contribution that Dr. Blum had made. Having known Dr. Blum personally, I was impressed with one thing and that was his concern for the success of the American Electroplaters' Society. A part of this concern was for harmony between the practical plater and the research man. This is well expressed in the first William Blum Lecture in which he concluded that "education in its broadest sense and application is the most important problem that confronts the plating industry." With this thought in mind, I thought that the subject of decorative coatings discussed from the practical and theoretical aspects would be apropos for this lecture.
History
History shows that bright finishes appealed to man in very early time. The Egyptian tombs have revealed many articles that were decorative in some way, partly as a result of their being smooth, bright and reflective. Of course, many of these items were solid gold and because of the nobility of the metal, they retained their bright finish. Today, the use of a solid noble metal would be prohibitive both from the standpoint of cost as well as availability. The result therefore has been to resort to plated coatings to conserve metal and reduce costs. However, there is still the demand for the durable, bright, decorative finish that we have come to associate with quality.
Background
Plated nickel coatings that were first used for their decorative appearance were dull as-plated and required buffing to bring up a lustrous finish. In the atmosphere, frequent cleaning and polishing were required to maintain the original appearance. The discovery that bright chromium could be plated over the buffed nickel provided a permanence to the finish. This combination of nickel and chromium performed well because the thick nickel layer provided good corrosion resistance and protection of the basis metal and the chromium coating provided a surface that was resistant to staining. Even though this was a big step forward at that time, the metal finishing industry was well aware that there was need for improvement. The objective of their research centered on a better performing and less expensive system.
The development of an improved system followed several lines of attack. Progress was made both from an empirical as well as a theoretical approach and in many instances the two were complimentary. At the outset, progress was hampered by the lack of a theory to account for a mechanism for the deterioration of a decorative nickel-chromium coating. It was also hampered by the lack of a reliable accelerated performance test to appraise and thus determine quickly the influence of a variable being studied. A third factor was a system for rating the performance of a plated coating so that objective comparisons could be made between two systems. All three of these problems have been dealt with, and the AES through its Research Program made a major contribution in the development of accelerated tests, the Corrodkote and CASS tests. The results of research on these problems have expedited the development decorative coatings to the point they are today.
Exposure tests
At first, static exposure of plated panels met the need for information on composition, thickness and other properties of the coating. One of the first comprehensive programs of this type was carried out under the direction of Dr. Blum at the National Bureau of Standards in cooperation with AES and ASTM. In this program in 1932, SAE 1010 steel was used for the basis material and Watts nickel was buffed and chromium plated. The conclusions from this exposure program were follows:1
- Thickness of nickel was the most important factor.
- Copper under the nickel was detrimental only with thin nickel coatings.
- Chromium coating resisted tarnish but added little to protective value.
- Pretreatment or plating conditions had no marked effect.
In a later program2 in 1936, zinc-base die castings and brass were included with several types of steel. Buffed Watts nickel was again the principal coating but a few bright nickels available at that time were included. The conclusions arrived at from this work were as follows:
- Thickness of nickel was the most important factor on all three basis materials.
- Copper added little to protective value on steel or zinc.
- Type of nickel had no large effect but there was more variability with the bright nickels.
- Optimum thickness of chromium appeared to be in the range of 0.25 to 0.75 μm (10-30 microinches) above which cracking occurred.
The variables studied in these programs and the results obtained raised many questions to be answered. Among the major ones were the cause of pitting and corrosion, effect of basis metal, effect of type of nickel, effect of copper under nickel and the effect of the chromium layer.
Nature of corrosion
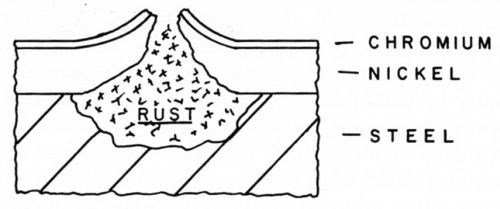
Corrosion of coatings manifested itself by severe pitting and general staining of the surface. In some cases the pitting was so severe that the surface took on the appearance of a "nutmeg grater" and has been classified as crater rusting in the new system of rating. At this stage of deterioration the surface could no longer be cleaned to restore the lustrous appearance. Examination of these pits in cross section, shown schematically in Fig. 1, established that corrosion had penetrated to the steel basis metal and that the expansive pressure of the corrosion products had raised the coating around the periphery of the pit. The exposure results indicated that this type of failure was more pronounced in salt-containing atmospheres and in the presence of a copper undercoat.
Figure 1 - Crater rust-type pit.
About this time, Dr. Wesley theorized that the corrosion products of copper could be responsible for the accelerating effect on the deterioration of copper-nickel-chromium coatings. It was known at that time that soluble salts of copper in acid solutions could accelerate the corrosion of nickel and other metals. Therefore in an atmosphere containing sulfur dioxide, copper undercoats might be expected to accelerate deterioration of nickel-chromium coatings. However, in an atmosphere which is neutral in reaction, the copper corrosion products would be insoluble and therefore without effect. In the same way, iron corrosion products might affect the deterioration of these coatings. This theory was substantiated by Knapp and Wesley3 by exposing nickel-chromium-plated steel panels so that the wash from copper and iron screens was allowed to drip across the plated surface as shown in Fig. 2. The results showed that both copper and iron corrosion products could accelerate the corrosion of nickel-chromium coatings in the atmosphere. Therefore the corrosion products of any exposed copper or iron on a plated part could accelerate the attack on the adjacent nickel.
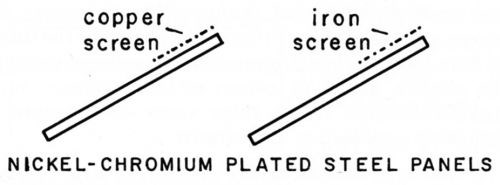
Figure 2 - Exposure test for effect of copper and iron corrosion products.
Other evidence of the effects of copper and iron salts was later revealed in the development of the Corrodkote test in AES Research Project 15.4 In this work, analysis of the Detroit atmosphere showed the presence of both copper and iron salts and, when they were incorporated in the test media, they did indeed accelerate the corrosion of nickel-chromium coatings. Copper salts were also found to have a similar effect in the solution used in the CASS accelerated test.5 The theoretical explanation for the behavior of copper and iron salts on the corrosion of nickel-chromium coatings is well described by Petrocelli, Hosparaduk and DiBari6 in their study of the CASS and Corrodkote electrolytes. Both salts were found to act as cathode depolarizers and thus increased the corrosion current in the pitting cells. Normally, one would expect the action of these salts to subside as a result of their being reduced in the cathode film from the cupric and ferric states. However, the conditions in the film of CASS solution and the moist Corrodkote paste provide access to atmospheric oxygen which oxidizes the copper and iron salts back to their higher valency states.
Effect of basis metal
Nickel-chromium coatings on steel performed reasonably well but rust spotting developed in time, the duration depending on type of exposure and thickness of coating. It was recognized that some of the pitting that developed these rust spots emanated from defects in the steel surface which in turn had influenced the continuity of the nickel layer. The importance of such a condition on the steel surface was described in a paper by Pinner.7 It was shown that defects generated from polishing a surface could lead to points of weakness in the resultant coating. A method was developed to eliminate this condition and thus insure improved polished steel surfaces for plating. It was also believed that the influence of some defects in the substrate surface could be minimized by first plating with copper, which allegedly has better covering power than nickel. This would also reduce the porosity of the subsequent nickel layer. Of course, there were other advantages to using copper prior to nickel - namely, it was more readily buffed to a high finish and it was cheaper than nickel. At that time, it was also favored because of the experience with copper-nickel composites on zinc.
Effect of copper undercoats
The question of the effect of copper undercoats on durability of nickel-chromium seemed to be a perennial one. At the close of World War II, ASTM Committee B8 in its Program No. 1 initiated an exposure program primarily to resolve this question.8 A few bright nickels were included in this program but the majority of the panels were coated with buffed Watts nickel. A notable variable in the program was a double-layer nickel system in which the first layer was buffed and then a second layer was applied and buffed. Although the exposures were more varied than those programs reported by the Bureau of Standards, the conclusions were the same pertaining to the relative merits of copper-nickel-chromium versus nickel-chromium. After three years of exposure the following conclusions were drawn:
- The type of atmosphere had a very large effect on the rate of deterioration.
- The thickness of nickel was the principal factor.
- Copper undercoats added little to protection and that was early in the exposure and in milder atmospheres.
- Type of nickel plate had no effect.
- Double-layer nickel was highly beneficial in marine atmosphere but not in industrial atmosphere.
The unusual finding in this program was the beneficial effect of double-layer nickel coatings. Was the structure of the second layer changed as a result of plating over the buffed first layer? Why didn't plating over buffed copper have a similar effect? These questions led to research on porosity and the cause of pitting.
Porosity theory
About this time, it was believed that the failure of nickel-chromium coatings was related to the inherent porosity of the nickel layer. It was also theorized that the corrosion followed the grain boundaries of the columnar-shaped grains of the Watts nickel. This would account for the rapid perforation of the nickel layer and the appearance of corrosion products of the basis metal. The ferroxyl test had been considered as a reliable method for determining the porosity of the nickel coatings. However, when it was tested on foils, it was found to actually create pores, as shown in Fig. 3. This test and other porosity tests, when examined, were found to be corrosive to nickel. The corrosiveness of the solutions used correlated with the number of perforations observed in the foils. In a study on the performance of nickel foils in the atmosphere, Wesley and Knapp9 concluded that nickel deposits are far less porous than originally thought and that corrosion pitting of the surface leading to perforation was the chief cause of failure.

Figure 3 - Nickel foil exposed to ferroxyl test solution.
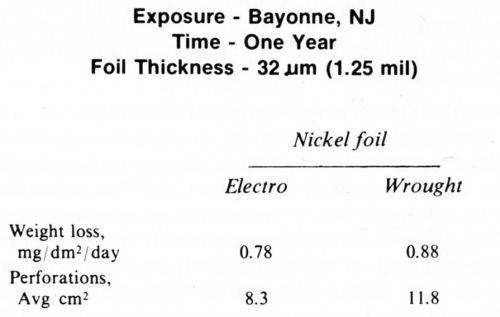
Electrodeposited nickel foils were exposed to the atmosphere along with wrought nickel foil of the same thickness. The behavior of the two types of foils was essentially the same with respect to both weight loss and number of perforations, as indicated in Table 1. This would suggest that the inherent porosity of the electrodeposited foil was insignificant. A chromium coating on these foils appeared to be beneficial in the early stages of exposure but in time it increased the number of perforations. Similar results with foils were obtained by Brown and Hoover.10
Table 1 - Atmospheric exposure of nickel foil.
Porosity of nickel foils was also the subject of AES Research Project 6. In this investigation, Dr. Thon found that the inherent porosity of electrodeposited nickel foil as determined by gas permeability was essentially zero, even at extremely thin thicknesses. These results were also confirmed by Ogburn at the Bureau of Standards in AES Research Project 13.
Corrosion pitting
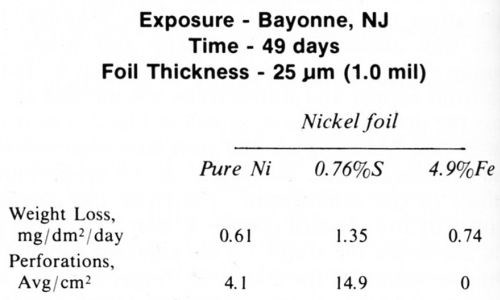
An empirical study by Wesley and Knapp9 investigated the effect of codeposited material in the nickel on the pitting tendency. Nickel plating baths were treated so that small amounts of lead, cobalt, iron, copper, sulfur and arsenic were codeposited with the nickel. The deposits were plated in the form of foils and exposed to an industrial atmosphere. The most striking results were those related to sulfur and iron in the deposit. The data in Table 2 show that sulfur increased the weight loss as well as the number of perforations, whereas iron increased the weight loss only slightly but eliminated the perforations. The finding that about five per cent iron in the nickel prevented perforations was most exciting, since the goal was to find some way to prevent corrosion pitting from reaching the basis metal.
Table 2 - Atmospheric exposure of nickel foil containing sulfur and iron.
In the next series of tests, chromium plate was tested on these foils. Again, the iron-containing foil resisted perforation but it became stained with rust very quickly. After one year of exposure, however, the iron-containing foil had many more perforations than pure nickel, even though they both had about the same weight loss. A similar observation was made by Brown and Hoover.11 This is interesting in view of the fact that years later, nickel-iron alloy deposits are being used in decorative plating. Exposure tests on these coatings by Clauss11 confirm these results that the alloy in the atmosphere becomes stained quickly with iron rust when chromium plated. An intermediate layer of bright nickel deters the appearance of rust stain. At a recent symposium, it was noted that because of this staining problem, nickel-iron electrodeposits are not recommended for exterior applications.
The effect of an overplate of chromium on the nickel foils was surprising. Early in their exposure, chromium reduced the number of perforations and weight loss but on prolonged exposure it caused a rapid increase in the number of perforations, although weight loss remained constant. This was true for both sulfur-free and sulfur-containing nickel but the perforation rate was higher for the sulfur-containing nickel. This observation was contrary to that made by Sample,12 where he reported that bright nickel on steel developed fewer rust spots than Watts nickel and an overplate of chromium reversed this performance.
To learn more about the factors that affect corrosion pitting of nickel-chromium coatings, Knapp13 prepared composite coatings in which a second metal was interposed between two layers of nickel. The metals were selected to represent a wide range of electrochemical activity from strongly active to extremely passive metals. Watts nickel on steel and as foils were used in the first program. Blum and Strausser2 had previously tested some sandwich coatings of nickel-copper-nickel and found that they were inferior to the same thickness of nickel. This is understandable from the previous work on effect of copper corrosion products that act as a cathode depolarizer and accelerate the pitting corrosion. In this series of tests, cobalt and chromium as the intermediate layer produced the most pronounced effect in delaying perforation of the coating. The mechanism by which these two metals delayed perforation was different. Cobalt acted as an anode when it was exposed in the pit and corroded sacrificially to protect the nickel. This composite with cobalt tended to form blisters as shown in the schematic profile of a pit, Fig. 4. The surface area affected was small because the pit opening was cathodically protected. Chromium had the opposite effect because it was cathodic to nickel. This was evident from the profile of the pit which was large and flat-bottomed as shown schematically in Fig. 5. The pitting of the outer layer proceeded until the chromium was exposed and then the corrosion of the nickel spread laterally. The thickness of the chromium was not critical and a conventional decorative thickness was adequate. Of these two metals, chromium is best suited from the practical standpoint. A special step is required to plate nickel adherently on the chromium and, because of this and other attendant problems, no practical application was ever made of this system. These results with chromium were confirmed by other investigators on zinc and in other atmospheres.14,15
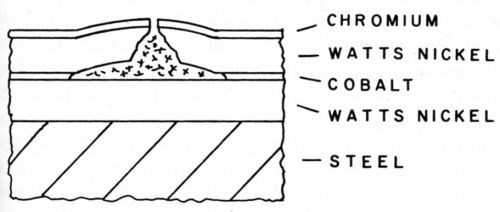
Figure 4 - Pit in nickel composite with cobalt.
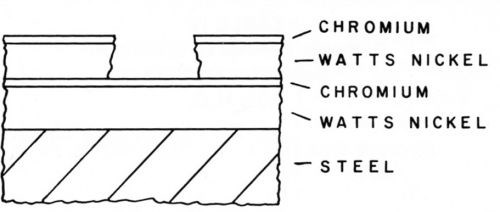
Figure 5 - Pit in nickel composite with chromium.
Bright nickel
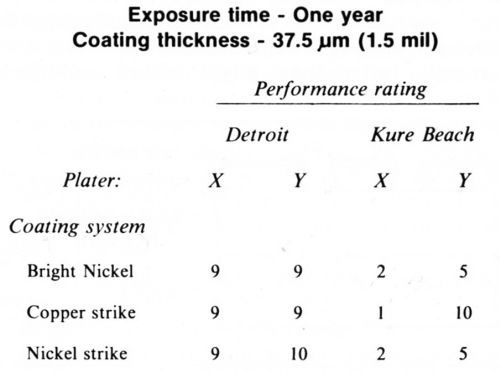
The research and development of bright nickel plating systems is a story in itself and has been well covered by Dr. Brown in his William Blum Lecture.16 Bright nickel baths were continually improved starting in 1934 and were used extensively in the auto industry for plating on both steel and zinc die castings. In 1961, ASTM Committee B8 reported on an extensive program designed to determine influence of some plating variables on the performance of bright nickel coatings.8 Several variations of the use of nickel and copper strikes under the bright nickel were investigated. Steel panels plated primarily with a single-layer bright nickel were prepared under identical standardized conditions by two platers (designated by X and Y). Exposures were made in Detroit and Kure Beach. Good agreement was obtained between platers X and Y on all panels at Detroit and on all coatings except those containing copper at Kure Beach. As shown in Table 3, the panels with the copper-nickel system plated by Y were distinctly superior. The committee reported that they were unable to find a reason for this discrepancy.
Table 3 - ASTM exposure tests of nickel-chromium coatings.
Semi-bright nickel
About this time, the auto industry was becoming dissatisfied with the performance of bright nickel in acidic industrial atmospheres or in areas where salt was used to de-ice roads. They demanded a system that performed more like Watts nickel but yet was economical. An answer to this demand came when DuRose reported in 1945 on the use of coumarin in a nickel plating bath.17 This had a leveling action on the deposit and thus produced a semi-bright appearance. The deposits could be easily buffed to a bright finish and thus they were accepted as having a real economic advantage. They were also sulfur-free, so they had the good performance properties of Watts nickel. The deposits were also ductile and were thus better suited than bright nickel for such applications as automobile bumpers. Data from an exposure program on plated panels reported by Brown and Millage18 indicated that the semi-bright nickel and Watts nickel were comparable and superior to bright nickel coatings.
Duplex nickel
The economic stimulus was to reduce costs even further and the next step eliminated buffing of the semi-bright nickel by plating bright nickel directly over it. This double-layer nickel coating known as "duplex nickel" was found to be superior to even the double-layer Watts nickel, which of course was not economical and thus never received extensive application. DuRose and Pierce19 reported on tests of the double-layer nickel in a number of atmospheric exposures and found that it was consistently better than bright nickel coatings of comparable thickness. Examination of corrosion pits in the double-layer coatings revealed that the difference in activity of the two layers was responsible for the improved performance. It has been well documented by several investigators that the appearance of a pit in the double layer is as shown schematically in Fig. 6. The mechanism is that the pitting action proceeds through the bright nickel but stops at the semi-bright nickel interface and then proceeds laterally in the bright nickel layer. Several investigators20 have shown that the sulfur in the nickel does indeed make it more active electrochemically. In combination with the sulfur-free nickel it acts to sacrificially protect it. It has been noted, however, in atmospheric tests that the protective effect is reduced in industrial atmospheres. Laboratory research has shown that sulfur dioxide in the atmosphere activates the sulfur-free nickel in the same way as sulfur in the bright nickel.
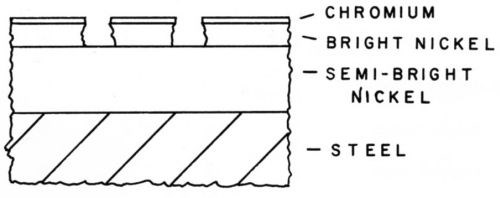
Figure 6 - Pits in duplex nixkel.
This duplex nickel coating represented a major step forward in obtaining improved performance of decorative coatings on both steel and zinc. As a result they have been used extensively and their performance in most cases has been rated good.
A modification of the double layer was made by Brown,21 who introduced a third layer of nickel between the semi-bright and bright nickel. This layer was thin but was so significantly higher in sulfur that it gave additional cathodic protection to the other nickel layers and thus further delayed perforation of the coating. The pitting action is shown schematically in Fig. 7.
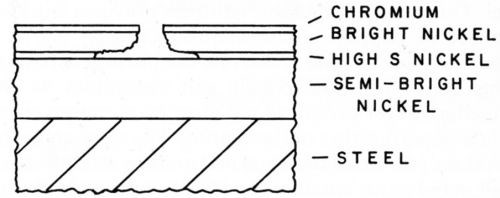
Figure 7 - Pit in tri-Ni system.
Chromium
Despite the generally good performance of the duplex nickel coatings with regular chromium it was recognized that the appearance could be improved. In atmospheres or conditions where surface pitting was severe, the pits were broad (Fig. 6) and this detracted from the lustrous appearance of the coating. There had been speculation that the chromium coating itself could influence the nature of the pitting. The pitting theory of corrosion of nickel-chromium coatings would predict that the density of pores in the chromium could influence their performance. Numerous investigations had established that regular chromium coatings were porous below 0.25 μm (10 microinches) and above 0.5 μm (20 microinches) and tended to form craze cracks. A thick chromium layer free of pores and cracks would appear to be an ideal coating over the bright nickel. Considerable research was devoted to this objective of plating thicker chromium that was free of cracks. Several investigators22 found that by altering the chromium plating conditions and/or catalyst in the bath, the thicker deposits obtained performed better in both accelerated and atmospheric tests. The principal disadvantages of this system were the longer plating times and the fact that many of the thicker deposits required buffing to produce the desired luster. ASTM exposed a program of plated panels in which several types and thicknesses of chromium were used over the double-layer nickel on steel. In both marine and industrial locations, chromium deposits thicker than 0.25 μm (10 microinches) gave better protection. However, the thicker chromium deposits developed a craze pattern that detracted from the appearance.
Double layer chromium
Double-layer chromium was developed to retain the benefits of crack-free chromium and the appearance of regular chromium. This was accomplished by plating a layer of crack-free chromium on the bright nickel followed by a layer of bright and finely cracked chromium. Exposure tests and commercial experience indicated that this combination gave superior performance as compared to regular chromium. Again the drawbacks to this system were the lack of luster as produced and the development of haziness on exposure.
Microcracked chromium
The high stress in the thick chromium plates of the double-layer chromium system indicated that the cracks were penetrating to the nickel layer and that a marked increase in corrosion protection was obtained by going in the opposite direction to the crack-free system. The incentive then turned to producing a highly cracked chromium layer. Several observations in research on the pitting theory of corrosion indicated that the nickel-chromium couple was under cathodic control. Thomas23 found that when different ratios of nickel and chromium were exposed to a CASS electrolyte, the amount of nickel corroded was the same when the area of chromium was constant. Lovell24 demonstrated this in an experiment and obtained the data in Table 4. With equal areas of nickel and chromium the cell produced a current of 100 μA. Reduction of the chromium area by half reduced the current in half. However, with reduced areas of nickel the current remained constant. This explains the effect of a few pores in regular chromium versus many pores or cracks in microcracked chromium. The corrosion rate of the nickel at a few pores would be high, leading to rapid penetration, whereas at many cracks it would be low and thus the penetration rate would be low. A schematic of the pitting in microcracked chromium is shown in Fig. 8.
Table 4 - Cathodic control of chromium solution - 1N NaCl, 0.3% H2O2.

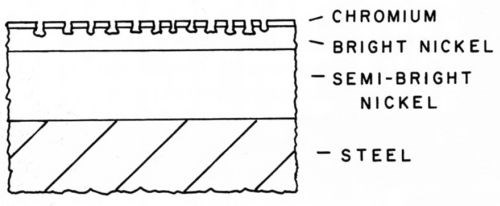
Figure 8 - Pits in microcracked chromium.
A second method for producing a microcracked chromium was developed by plating a very thin, highly stressed layer of nickel on the bright nickel. The nickel was plated from an all-chloride bath to give the high stress and it was plated sufficiently thin that it did not affect the brightness. When this surface was then plated with the highly stressed regular chromium, a fine microcrack pattern was produced in the chromium layer.
Microporous chromium
A related research development that led to a different form of high porosity in the chromium was satin nickel. The original incentive here was to produce a complimentary finish to the lustrous bright nickel by some means other than a mechanical one. A process was developed whereby a satin finish could be obtained directly in a plating bath by codepositing fine non-metallic particles dispersed in the nickel bath. The size of the particle and the type of bath controlled the degree of the satin finish. When these coatings were chromium plated and exposed in the atmosphere, they were found to have outstanding protection to the basis metal. This led to the development of the microporous chromium system,25 which operates under the same principle as the microcracked system.
Another method of producing a highly porous chromium layer is to subject the regular chromium deposit to impingement of fine particles. This system is called the "Pixie" process.26 The principle of this process finally explained the results that Sample obtained when he compared the performance of nickel-chromium-plated panels at Kure Beach in the 80- and 800-ft exposure. Surprisingly, he found that the panels exposed only 80 ft from the surf were outstanding in their performance compared to those at 800 ft. Impingement of the surface by the blowing sand had created the "Pixie" effect.
Diffused chromium
A unique system for coating a steel bumper with chromium was developed and patented in 1964.27 It was a high-temperature diffusion process in a molten salt bath and it did not involve electroplating. Fortunately, for the plating industry, it was not completely successful but it did demonstrate that it was possible to produce a pore-free, ductile and stress-free coating of chromium.
Present status
The ultimate, then, in the present state of the "art" for a good, durable, decorative coating is a microporous or microcracked chromium plate over a duplex nickel. The still controversial question is whether a copper layer under the nickel is beneficial.28 There seem to be two schools of thought on this and both have data to support their contention. One is that copper acts as a barrier layer and is protected cathodically by the sulfur-containing bright nickel in the coating. The other is that the copper corrosion products accelerate the pitting corrosion. Apparently only more research and testing will resolve this question.
Prognostications
After a review of decorative coatings one cannot resist speculating as to what future developments might be. I believe that the opinion is generally accepted that the lustrous finish obtained with a nickel chromium system with or without copper cannot be surpassed economically by any other method. So where should one look for improvement? Perhaps better performance at less cost? What would the qualifications be of an ideal decorative coating?
- Leveling deposit - to eliminate mechanical finishing.
- Brightness or luster - for eye appeal.
- Ductility - to withstand impacts and deformation.
- Performance - to retain the initial appearance.
- Suitable for all substrates - steel, zinc, aluminum and plastics.
Of these qualifications where is the greatest opportunity for improvement? My answer is brightness and performance because in the best system today they are interrelated. We do not have full brightness with the microporous or microcracked chromium system and we do not have good performance with the full bright system with regular chromium. Wouldn't we have a perfect system if we could just change the properties of the chromium layer to a pore-free, ductile and stress-free deposit? I think that is the goal!
References
1. W. Blum, P.W.C. Strausser and A. Brenner, J. Res. Nat. Bur. Standards, 13, 331 (1934).
2. W. Blum and P.W.C. Strausser, ibid., 24, 443 (1940).
3. B.B. Knapp and W.A. Wesley, Plating, 38, 36 (January 1951).
4. D.M. Bigge, Proc. Amer. Electroplaters' Soc., 46, 149 (1959).
5. C.F. Nixon, J.D. Thomas and D.W. Hardesty, ibid., 46, 159 (1959).
6. J.V. Petrocelli, V. Hospadaruk and G.A. DiBari, Plating, 49, 50 (January 1962).
7. W.L. Pinner, ibid., 40, 115, (October 1953).
8. Report of ASTM Committee B8, Proc. ASTM, 53, 256 (1953); Report of ASTM Committee B8, ibid., 61, 326 (1961).
9. W.A. Wesley and B.B. Knapp, Trans. Inst. Met. Finishing, 31, 267 (1954).
10. H. Brown and E.W. Hoover, Proc. Amer. Electroplaters' Soc., 40, 32 (1953).
11. R.J. Clauss and R.A. Tremmel, Plating, 60, 803 (August 1973).
12. C.H. Sample, Plating, 47, 297 (1960).
13. B.B. Knapp, Trans. Inst. Met. Finishing, 35, 139 (1958).
14. W.H. Safranek, H.R. Miller and C.L. Faust, Proc. Amer. Electroplaters' Soc., 46, 133 (1959).
15. H. Brown and M. Weinberg, ibid., 46, 128 (1959).
16. H. Brown, Plating, 55, 1047 (October 1968).
17. A.H. DuRose, U. S. patent 2,635,076 (1953).
18. H. Brown and D.R. Millage, Trans. Inst. Met. Finishing, 37, 21 (1960).
19. A.H. DuRose and W.J. Pierce, Metal Finishing, 57, 44 (1959).
20. V. Hospadaruk and J.V. Petrocelli, Plating, 48, 479 (May 1961).
21. H. Brown, Metalloberflache, 11, 333 (1962).
22. R. Dow and J.E. Stareck, Proc. Amer. Electroplaters' Soc., 40, 53 (1953); H. Brown, M. Weinberg and R.J. Clauss, Plating, 45, 144 (February 1958); E.J. Seyb and W.H. Rowan, ibid., 46, 144 (February 1959).
23. J.D. Thomas, D.W. Hardesty and C.F. Nixon, Proc. Amer. Electroplaters' Soc., 47, 90 (1960).
24. W.E. Lovell, E.H. Shotwell and J. Boyd, ibid., 47, 215 (1960).
25. T.W. Tomaszewski, R.J. Clauss and H. Brown, ibid., 50, 169 (1963).
26. T. Malak, D. Snyder and A.H. DuRose, Plating, 59, 659 (July 1972).
28. A.H. DuRose, ibid., 62, 941 (October 1975).
27. British patent 964,323 (1964).
About the author:
This piece was written at the time Dr. Knapp was announced as the recipient of the 1977 Scientific Achievement Award:

Born near Northfield, Minnesota on May 29, 1910, Dr. Knapp attended public schools there and obtained a B.A. degree in chemistry from Carleton College. Following graduation, he was granted a research assistantship in the chemistry department at the University of Wisconsin, where he had the opportunity to study under O. P. Watts, a pioneer in nickel plating.
In 1937 Dr. Knapp earned his Ph.D. and from that time on was associated with the International Nickel Company. While attending the summer meeting of the American Chemical Society, he met Dr. W.A. Wesley of INCO, and, as a result of that meeting, became employed in the company's laboratory in Bayonne, New Jersey. He retired two years ago as Research Supervisor of INCO's Electrochemical Section at the Paul D. Merica Research Laboratory in Sterling Forest, Suffern, New York.
Dr. Knapp has specialized in the field of corrosion and electroplating - particularly nickel plating. A major contribution of his, summarized in the paper "Weathering of Nickel-Chromium Electrodeposits," helped to establish the multilayer coating concept and provide the basis for the use of double-layer nickel coating, now standard procedure in the industry. Experts in the field have credited Dr. Knapp's advice and consultation as important aspects in the area of decorative nickel-chromium coatings. His research in this area brought about a breakthrough which lead to the conclusion that the failure of coatings was due to pitting corrosion rather than porosity of the nickel layer.
He is also the co-developer of SD-Nickel, an active form of electrolytic nickel used widely in titanium anode baskets. More recently he was instrumental in developing a new form of the material S-Nickel Rounds, made by an electrorefining technique that he developed in cooperation with production facilities.
Dr. Knapp holds many patents, the first of which was granted in 1941. A few of his patent titles are: "Black Finishes on Nickel Surfaces," "Black Nickel Plating," "Auxiliary Nickel Anode," "Bath for Depositing Hard Nickel Plate" and "Insoluble Anodes for Nickel Plating Baths."
Among the numerous papers he has authored or co-authored, "Notes on Nickel Sulfamate Plating" was particularly helpful to electroformers in understanding the operation of nickel sulfamate plating baths. The work also helped to establish methods of controlling the properties of deposits from nickel sulfamate baths.
Dr. Knapp has also invested considerable energy in many organizations and societies. He has been an active member of the AES Newark Branch for more than 25 years, and has held several officer's posts at the national level. These include Research Board Chairman, member of the Technical Education Board, Chairman of the Paper Awards Committee, and Chairman of the Scientific Achievement Award Committee. He is a long-standing member of the ASTM Committee B08, in which he has been helpful in designing and conducting panel exposure programs and assessing the performance of coatings on steel, zinc and other materials. His efforts have helped ASTM establish specifications to help assure the quality of decorative coatings. He is also a member of the American Chemical Society, the Institute of Metal Finishing, the Electrochemical Society, Alpha Chi Sigma, Sigma Xi and RESA, and is listed in the American Men of Science.
RELATED CONTENT
-
Electroless Nickel-plated Steel versus Stainless Steel: Case Studies
This paper highlights two case studies of manufacturers that have replaced, or done studies to replace, stainless steel with electroless nickel-plated mild steel. In both cases, cost savings could be realized while maintaining or improving product quality.
-
Chromium-free Etching and Palladium-free Plating of Plastics
Plastics are replacing metals in the manufacture of many parts, and quite often there is a need for metallic coatings on the plastics and other non-conductors. This paper will describe new processes of preparing ABS plastic substrates for subsequent metallization.
-
History of Chromium Plating
A review article originally printed as Plating & Surface Finishing, 71 (6), 84-91 (1984) on the occasion of the 75th anniversary of the founding of the American Electroplaters Society.