Solving Adhesion Issues from Laser Cuts
Remove scale with a pretreatment solution.
Using lasers to cut steel is becoming more common today because they provide an increase in finished part quality and a reduction in manufacturing costs.
The downside, however, is reduced paint adhesion on cut edges because a poorly-adherent iron oxide scale is formed due to the reaction of the molten steel with oxygen. When this loose scale is painted over, adhesion failures can often occur upon impact because the paint adheres to the scale, but the scale doesn’t adhere to the steel. The best way to avoid this is to remove the laser scale before painting.
Featured Content
2 Ways to Rid Scale
Mechanical methods of removing scale—such as grinding or abrasive blasting—are simple, but often not effective on intricate cuts, which is one of the reasons for the use of a laser in the first place.
The other method of scale removal is acid pickling. There are a wide variety of acids that can be used to remove scale, but not all are practical for use in conventional paint pretreatment processes. For years, phosphoric acid was the most commonly used acid for removal of laser scale and other oxides from steel in pretreatment processes. One of its chief advantages is that drag-out of phosphoric acid into a phosphate conversion coating will have minimal effect on the phosphate bath chemistry.
Phosphoric acid is generally more expensive than many other acids, however, and when used on steel will generate a considerable amount of sludge in the bath and scale on the heater surfaces.
With the increasing restrictions on phosphorous discharge, and the advent of phosphorous-free advanced pretreatments, the use of phosphoric acid pickles has fallen in recent years. Alternatives such as sulfuric acid-based products are becoming more common in prepaint processes. Sulfuric acid is inexpensive and low sludging and scaling. However, it can leave the steel more prone to rusting in the rinse than phosphoric acid and must not be dragged into the pretreatment bath where it can degrade paint performance. With good control of the rinsing stages however, sulfuric acid pickling is an excellent alternative to phosphoric and other acids.
Organic acids such as citric acid have the advantages of being less expensive than phosphoric acid, and because they are sequestrants, they keep the dissolved iron in solution thereby preventing sludge and scale formation. However, organic acids can interfere with precipitation waste treatment systems, so if there are other metals such as zinc being treated in the waste treatment system, it may be necessary to segregate the organic acid from the zinc-containing waste stream.
Finally, mixtures of acids sometimes combined with surfactants and inhibitors are commonly used in pretreatment processes. Mixtures can be formulated to give the advantages of certain acids while minimizing the disadvantages of others.
Best Parameters
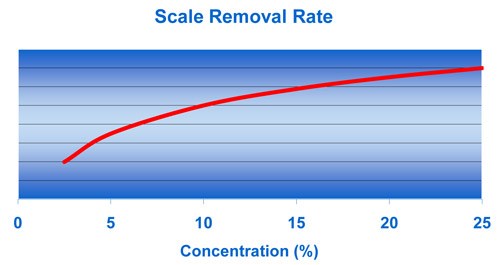
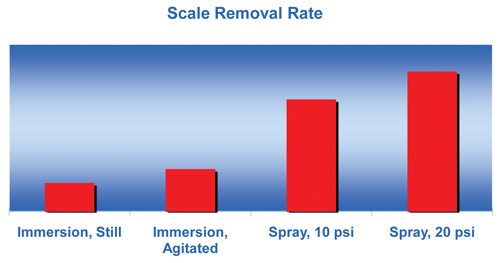
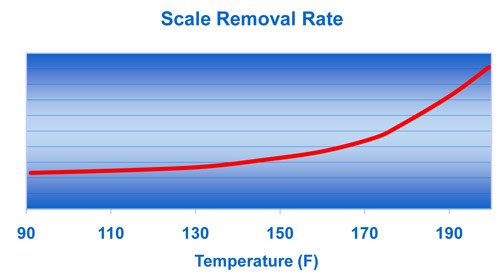
The scale removal rate is dependent on the acid concentration, temperature, rate of solution movement across the metal and the amount of dissolved iron. These charts show that, although acid concentration is important, the acid temperature and degree of agitation (or spray pressure) are the more effective variables in increasing the scale removal rate.
For example, Figure 2 shows an increase in the scale removal rate as concentration is increased, but there is a flattening of the curve past 20% concentration. Figure 3 shows that a high degree of solution movement across the metal, especially with spray, results in a dramatic increase in scale removal.
Figure 4 shows that as the temperature is increased, there is an exponential increase in the scale removal rate. The conclusion with regard to parameters is that higher temperature and solution movement have the most impact on the scale removal rate.
As the bath is used, dissolved ferrous iron builds in solution. With this, the pickling rate will gradually decrease as the iron content increases.
What About Rinsing?
Acid pickled steel is highly active and prone to rusting. An effective method to avoid this is to use a slightly alkaline neutralizing rinse. The pH should be controlled at about 8 to 9 by adding an alkaline material such as caustic, soda ash or other inhibiting alkaline materials.
To minimize the effect of drag-out of alkaline neutralizing material, a second water rinse, or exit fresh water halo rinse can be used. The most effective and economical means of controlling a neutralizing rinse is with a simple automatic pH controller. Controllers are available which continuously monitor the rinse pH and feed the neutralizer using an integrated feed pump to keep the pH at the pre-set value.
Waste Treatment
The decision whether to waste treat used acid pickle baths in-house or have them hauled away must be made based on the capability of the waste treatment system, the constituents of the used solution, and the local discharge limits. Used pickle baths may contain organic soils, surfactants, inhibitors, and low levels of chromium, nickel, molybdenum and other metals dissolved from the pickled steel and from stainless steel equipment exposed to the acid.
Materials of Construction
For typical prepaint acid pickling processes using phosphoric acid, organic acids or mixtures, and under controlled parameters, sulfuric acid, stainless steel alloy 316L tanks and equipment are usually used. Heating surfaces, piping, pumps and valves are also usually constructed of stainless steel alloy 316L. It is important to use low-carbon grade (“L”) for both the sheet and the welding material.
Cast pump housings and valve bodies are usually constructed of CF8M which is an acid-resistant stainless alloy. For most acids, polypropylene or CPVC risers are suitable. Nozzles are often made of PVC or CPVC. Plastic and elastomer parts for gaskets, seals, etc.are usually made of PTFE, PVDF (Kynar) and FKM (Viton).
OSHA and other applicable regulations should be followed to ensure proper ventilation and to determine if a scrubber or mist eliminator is needed to remove fumes and/or mists. When an acid pickle is used in a stage other than the first stage, spray from the adjacent stages tends to remove acid mist.
Process Sequence
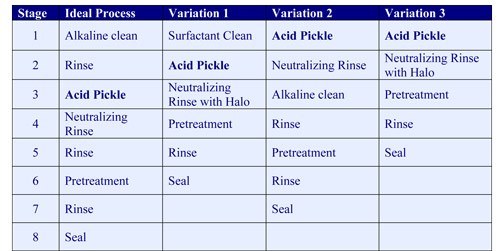
The first step is to remove organic soils such as mill oils, shop soils and metalworking fluids. This is best accomplished by using an alkaline cleaner followed by an overflowing water rinse (Ideal Process in the table on page 40). It’s important to remove organics first since acid pickles are not highly effective in removing these contaminants.
If the use of floor space or capital investment must be limited, it is possible to use a neutral cleaner such as a surfactant in the first stage followed by the acid pickle (Variation 1). In this sequence, the surfactant stage will remove light organic soils.
If not all the work is in need of acid pickling, it is possible to use an acid pickle and neutralizing rinse prior to a conventional five-stage process (Variation 2). With this sequence, the acid pickle can be operated part time, only for the work that has scale or rust, saving operating expense.
In cases where organic contaminants are light, it is possible to place an acid pickle in the first stage (Variation 3). This type of acid pickle should be formulated with surfactants to help remove the light organic soils and to keep them from redepositing on the work.
Once the organics are removed, the inorganics such as laser scale and rust are removed in the acid pickle stage.
After pickling, a neutralizing rinse is used to remove the acid and prevent rusting. After a neutralizing rinse, and an optional second fresh water rinse or halo, the work is ready for coating.
Bath Life
As the acid pickle is used, dissolved iron begins to build in the solution. As the iron content rises, the pickling effectiveness gradually falls. At some point, the pickling effectiveness drops to the point at which scale removal is ineffective. Sometimes the solution temperature can be increased in order to extend its life. With a typical prepaint acid pickling solution, a ferrous iron level somewhere on the order of 5 to 10 g/l will often be the maximum level tolerable. This can vary widely on individual processes.
With certain acids, such as phosphoric, it’s possible to achieve solution life of several years through the use of acid regeneration equipment, such as ion exchange.
Laser cutting can improve quality and cut costs, but the result is scale on the edges of the steel which can often cause poor paint adhesion. Acid pickling when incorporated into the pretreatment process is an effective solution. When designing a laser scale removal process, work closely with your equipment and chemical supplier.
Gary Nelson is surface treatment product manager at Chemetall US, Inc. For information, call 800-526-4473, or visit chemetallamericas.com.
RELATED CONTENT
-
Powder Coat MDF for an Enviable Finished Product
Cabinet maker says powder coating on wood offers more benefits.
-
Cleaning Prior to PVD/CVD Coating
Determining the cleanliness and chemical de-coating of PVD/CVD layers.
-
Alkaline Cleaning Guide
Gregg Sanko, Senior Chemist, Oakite Products, Inc. provides an overview of the alkaline cleaning process.