Specifying Buffing Compound
Buffing is still regarded by many in the industry as being something of an art form. In today’s competitive industry, where customers demand precision and reproducible results, that mindset is a dangerous one. The shift of buffing—in our collective consciousness—from art to science might be facilitated by a better comprehension of buffing compound.
"What is the right buffing compound to use for my application?" On the surface this appears to be a very simple question, but underneath the surface are lurking four more difficult questions to answer. Usually the real question that is being asked is:
What is the right buffing compound to use for my application that will...
Featured Content
- Remove all my scratches, handling marks and other imperfections,
- Bring out the natural luster of my metal,
- Easily clean off my part after buffing,
- Not add much cost to my process?
When (or should we say "if") these questions are finally answered and the perfect buffing compound is found, the inevitable next question is almost always, "Now how do I continue to get this same finish day after day, month after month, year after year?"
These are great questions to ask but our industry has not historically done a great job in answering them. All to often the usual answers are things like "It is a bit of an art" or "It is not an exact science." If we are to compete in a competitive world market, if we are to regain our place in the world as leaders in metal finishing, we must answer these questions better. We must move the process of buffing from the realm of Art into the realm of Science. In order to do that, we must first understand buffing compound better.
What is Buffing Compound?
There are three basic components to liquid buffing compound: grain, binders and water. The grain is what produces the desired finish on the part and without it there would be virtually no refinement in the surface roughness of the metal. There are primarily two types of grain used in buffing compound: Alumina is used in compounds to buff ferrous metals (steel, stainless steel, etc.) and Tripoli is used in compounds for nonferrous metals (brass, zinc, aluminum, etc.) A third grain, silica, is sometimes added to very fine (i.e., small grain size) Tripoli or Alumina to make color compound that can be used on either ferrous or nonferrous materials. Other grains may also be used but are less common for industrial use.
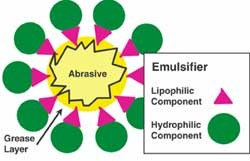
The grain must be "carried" to the buffing wheel where it can work on the metal. For liquid compound, water usually serves as the carrier. Unfortunately, if you mix any of the grains in with water, the grains will only sink to the bottom making it useless for any automated buffing system. Therefore, something else is needed to keep the grain suspended in the water. This is one of the jobs for the binder system in the liquid compound.
The binder system of most buffing compounds is composed of three items:
- Fatty Acids—aids in the buffing
- Petroleum Wax—adds tackiness to the buffing compound
- Amine or another emulsifying agent— helps form the soap with the acid which then sets-up the compound emulsion.
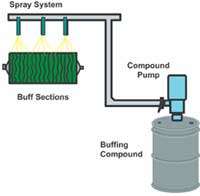
The emulsion keeps the grain/grease agglomerate in suspension in the water, enabling the compound to be easily transported with automatic systems to the compound guns where it is then sprayed on the wheel (Figure 2). The grease surrounds the grain producing the "tacky" feel that is necessary in most buffing compounds. This grease is what causes the compound to stick to the rotating buffing wheel where the job of finishing is accomplished. Unfortunately, it is the same greases that aid in "sticking" to the buffing wheel that also detracts by sticking to the metal workpiece after buffing. This issue is discussed later in the article.
Making the Part "Bright and Shiny": Cut and Color
The first underlying question was "How do I remove all my scratches, handling marks and other imperfections?" This is usually referred to as the cutting ability of the compound. This is often tied closely with the second question "How do I bring out the natural luster of my metal?" This is usually referred to as the coloring ability of the compound. But how do we know how much cut or how much color is required? For years, the industry has found a fast, accurate and reliable measure of surface finish to be elusive. Profilometers have been very good at measuring rougher surfaces such as that produced by abrasive belts or flap wheels. But a bright buffed finish can have the same Ra (3–4µm) as a dull finish because it is usually too low to be measured accurately.
Gloss meters have been much better at measuring a buffed surface but they have historically been very expensive and delicate. There have been some recent improvements in gloss meters to bring down the cost (often as low as $2,500–3,000) and making them smaller and more durable for use on the factory floor.
Once there is an accurate measure to define an acceptable finish, you can then vary the grain size and material to find the right balance between cut and color. The grain is the cutting tool that works on the part. Unlike other cutting tools, it is not held in a fixed position and this unique ability to "roll" when encountering an excessive torque produces the unique finish. The finish can vary depending on three characteristics of the compound:
- Grain Size
- Grain Shape
- Grain Hardness.
The grinding and screening process the grain goes through determines the grain size. The type of grain determines the shape. The type of grain also determines hardness, but for many grains the manufacturing process is a large factor as well.
Figure 3 provides a list of some of the more standard grains that are typically used in buffing compounds and the relative characteristics of each.
Cleaning the Part After Buffing
Are there characteristics that can make one compound formulation easier to clean than another? And if there are differences between compounds, are these characteristics measurable? The answer to both questions is "yes." Since the binder is what causes the compound to stick to the metal piece, then it reasons that if we can clean the binder then we can clean the compound residue.
| Figure 3: Standard Grains Used in Buffing Compounds | ||||
| Grain Chromium Oxide Iron Oxide 1 Micron Ground Alumina 5-7 Micron Ground Alumina 5-7 Micron Unground Alumina 9-12 Micron Unground Alumina Air Float Tripoli Double Ground Tripoli Once Ground Tripoli |
Grain Size Small Small Small Medium Medium Large Small Medium Large |
Shape Rounded Rounded Sharp Crystalline Sharp Crystalline Sharp Crystalline Sharp Crystalline Soft Crystalline Soft Crystalline Soft Crystalline |
Hardness Very Hard Very Hard Hard Hard Hard Hard Medium Medium Medium |
Type of Finish Produced Very High Color High Color Moderate to High Color Cut/Color Cut Aggressive Cut Moderate Color Cut/Color Cut/Color |
There are four aspects of the binder system that will be explained; with these four characteristics a compound can be more easily ranked by its relative "cleanability." There are other compound tests but the four discussed here are as follows:
- Saponification: This test measures the ability of the binder system to form an emulsion. An emulsion is formed when the saponifiable material is combined with an alkaline (this explains the emergence of alkaline cleaning systems). Once the binders/greases have been saponified, they can be easily removed with water.
One often-used test for saponification is AOCS (American Oil Chemists Society) method #Cd 3-25. This test calls for a strong base to saponify the binder at very high temperatures. It is a general test and probably needs further clarification to be of any help to most finishing operations. - Acid Value: This test measures the amount of free fatty acid in the binder system. The fatty acids in the compound will quickly and very easily form an emulsion in the presence of even a very mild alkaline solution. Therefore the higher the acid value the more readily the compound will clean from the part. It is a more specific test than saponification. The acid value of the binder in the buffing compounds can range from a high of 200 to around 30.
AOCS Test Method Te 1A-64 calls for the binder to be titrated with a mild alkaline solution. This test is highly accurate and repeatable. - Titer: This test measures the melting point of the binder system. This is especially important for the remaining unsaponifiable materials in the binder but both saponifiable and unsaponifiable binders must melt before they can be removed. Obviously, the lower the melting point, the more easily the residue could be cleaned from the part. Unfortunately, a balance must be found keeping the melt point low for cleaning and keeping it high for better tenacity to the buffing wheel. Average melting points will range from 110–160°F.
A standard test for Titer is AOCS 1A-64, which will provide the softening point for the binder system. - Iodine Value: This test measures the saturation of the carbon chain. A more saturated carbon chain results in a more stable compound. Unfortunately, it also causes more difficulty when trying to clean the compound. This test indicates the amount of double or triple bonds in the carbon chain. Under the right circumstances these types of bonds encourage the binder to form a soap, which can then be easily cleaned from the part. Since the iodine attaches where there is a double or triple carbon bond, the higher the iodine value, the easier it is to clean. Typical iodine values for compound are 1 to 10.
A popular test for Iodine Value is AOCS Cd 1-25, this will provide an accurate measurement of the saturation of the binder system.
Moving Forward
A thorough understanding of buffing compound is an important step in developing an approach to buffing that results in a more reproducible and cost-effective process. However, more needs to be done within the industry to improve the ability to control costs and quality of the surface finish. Finishers must continue to ask ourselves tough questions and not rest until we get answers that again make our products and processes the envy of the world.
Schaffner Mfg. Co., Inc.