Understanding Vibratory Finishing: Part 2 of 4: Media
The relationship of media, compound solutions, equipment and parts in successful vibratory finishing. how to improve your troubleshooting...
#basics
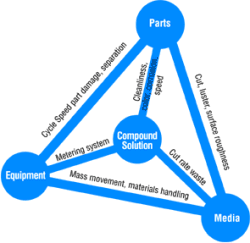
The media used in vibratory finishing was discussed in Part I. We showed that media, compound solution, parts and equipment are so interdependent that if one fails, the process fails. Now let's look at the compound solution and see how it relates to the other mass-finishing elements forming the "Tetrahedron of Interdependence" (Fig. 1).
The Compound-Water Solution
The correct use of compound-water solutions will be discussed in five sections: the compound solution and its functions; compound solution and media; compound solution and equipment; compound solution and parts; and compound solution and people.
Featured Content
The compound solution and its functions. As mentioned in Part I, the compound solution is the least understood and most frequently abused part of the mass finishing process. This is a good place to look first when trouble occurs.
Compound solution functions. The compound solution has the following functions:
- Control pH, foam and water hardness.
- Wet surfaces.
- Emulsify oily soils.
- Remove tarnish or scale.
- Control part color.
- Suspend soils.
- Control lubricity.
- Prevent corrosion.
- Provide cooling.
- Be environmentally compatible.
Control pH. The pH is a measure of the acidity or alkalinity of a water solution. A pH of 7 indicates that the solution is neutral-neither acidic nor basic. A pH of 1 or 2 indicates it is strongly acidic. High values--13 or 14--indicate it is strongly basic or alkaline. The pH is a log-scale measuring system. In other words, a solution of pH 3 is ten times as acidic as a solution of pH 4, and 100 times as strong as pH 5.
The pH of a solution is easy to measure by use of indicator paper or instruments. Solutions either very high or very low in pH are corrosive to people and dangerous to touch.
Do not expose the skin, eyes or mouth to such materials. Follow OSHA-20 form instructions carefully.
Many compounds are "buffered" to prevent big changes in pH from taking place. The term "pH control" may be a misnomer, since a good compound solution should take care of itself.
Mass finishing solutions vary from pH 1 to 14. While all of these have utility, the most popular ones are in the 4 to 14 range.
Control amount of foam. Some foam is necessary in most applications, especially those involving cleaning. Foam holds the solution in place on the surface of the parts and media. With no foam, process cycles are greatly extended or cleaning just doesn't occur.
Too much foam, on the other hand, kills the action in a vibratory machine. It cushions the mass so effectively that cutting can literally come to a stop. In rotary barrels or centrifugal barrels, however, which do not depend on energy transfer from the wall of the machine, foam merely moderates action. A barrel full to the top with foam is commonplace in burnishing or cleaning. Foam tempers the action.
Some plastic media generate considerable foam when run with plain water or non-defoaming compounds.
Oily soils can confuse the best of compound cleaners. These oily materials have the ability to defoam cleaning solutions, in effect removing the compound solution from the part it can no longer clean! Cleaning requires this contact of solution and soil.
Excessive foam is untidy, usually unnecessary and a morale reducer. Foam keeps mass finishing in the back of the plant, where nobody sees it. Result? No one cares. Costs, quality and personal satisfaction suffer.
Control water hardness. In the United States, water hardness varies from very low (one grain per gal or 17 ppm) to a high of over 30 grains per gallon or over 513 ppm. And public water supplies are often quite different from plant water coming from a well in the same area.
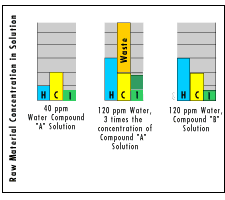
These great variations in hardness put a strain on the solution, for it must try to equalize the differences. In Fig. 2A, we see a perfect match between solution and water and process application. The raw materials are well suited to the needs of the process The solution of compound "A" in water is suited to the 40 ppm water supply, with just enough "H," the raw material used to take care of hardness. Raw materials "C," for cleaning and "1" for inhibitor are also used well.
Now the company changes to 120-ppm well water to reduce sewer backcharges. The solution now must be tripled in strength, as shown in Fig. 2B, to provide enough "H" for tripled hardness. But as noted, there is now a great excess of "C" and "I."
In Fig. 2C, a new compound was developed for the new application. All raw materials are again in balance. There is no waste. The product, compound "B," is more economical to use.
While slightly idealized, this concept is valid. It also helps explain why manufacturers of mass finishing compounds have so many from which to choose.
Wet surfaces. If the compound solution won't "wet" or spread on the surface to be cleaned, it won't clean well.
Take a drop of the solution at its use concentration. Put it on the surface to be cleaned. If it beads up like the water should on the hood of your waxed car, it is not wetting the surface. It cannot clean it. On the other hand, if it flows out and spreads on the surface, it has a chance to clean that surface.
As we'll see below, wetting is very dependent on concentration. Be certain, always, to check concentration first.
Emulsify oily soils. Emulsification is the process of surrounding very small droplets of oil with a surfactant so that the oil-loving end of the surfactant sticks into the oil, and the water-loving end sticks into the water. This holds the oil in the water and allows the solution to carry the oil along. The emulsion so formed can be stable for long periods or only for very short periods.
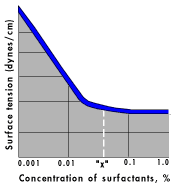
Fig. 4 shows the typical curve when surface tension of a surfactant is plotted against concentration. This curve tells us that at some point (shown by the arrow), activity of the surfactant is maximum--it can function at its best. This "threshold" concentration is necessary for cleaning. Even at slightly below this value we may get no cleaning at all.
When oily soils are emulsified, they don't easily deposit on other surfaces such as the lining of the machine or on the media. A dark band just above the load level in your vibrator means the soil is redepositing on the lining. You can bet cleaning is only marginal in this case.
Physical action improves the speed and stability of emulsification. Because vibratory finishers are excellent "scrubbers," they clean well. Media continually works the soils, allowing sometimes marginal cleaners to "get away with it."
Fig. 2 showed us the effect of tripling water hardness. If we increase the soil to be removed, the effect is similar. There wouldn't be enough "C" to emulsify the excess soil. Makes good sense to keep excess oils and greases out of the process, right? Saves money, too!
Remove tarnish or scale. Both high- and low-pH compound solutions can chemically attack tarnish and scales to remove them. These oxides of the metals darken the surfaces and lower reflectivity.
Highly alkaline solutions remove rust or scale from some steel surfaces. These alkaline solutions can produce very light, bleached metal surfaces. Strongly acidic solutions vigorously attack rust and other metal oxides. Unfortunately, they also attack the basis metal unless the solution is properly inhibited.
Suspend soils. Soil removed or made during the process can redeposit on the parts, on the media and/or on the lining of the chamber. This is especially troublesome when the media can't reach all surfaces of the parts. It is also detrimental in long cycles and when certain aluminum alloys are processed with plastic media. Excessively hard water contributes to the problem. How to overcome it? Tell your supplier. He should have a compound that prevents this.
Control lubricity. For years it was thought that to burnish or brighten a part, the solution had to be slippery. This is often the case in rotary barrels. In vibratory equipment, however, excessive slip brings everything to a halt. Traction is lost. Media stops rolling and nothing happens to the parts. Remember, these are very stupid machines. When roll slows or stops, so does work. It must!
One should not eliminate lubricity altogether, however. Just as some oil promotes cut of a machine tool bit, some lubricity in the compound solution assists cut of the so-called "deburring" compounds. Too much lubricity stops cut, but small amounts improve it.
Control part color. Coloring or darkening of parts in vibratory finishing is caused by oxidation or dirt impregnation. The lighter the surface, the cleaner it is. The highly alkaline compounds "bleach out" surfaces and sometimes give richer appearances. Acidic compounds sometimes give too clean a surface.
Inhibit corrosion. In a process that uses water, mixes it intimately with air, and then places this mixture on the surface of chemically clean metals, corrosion must occur. The only way to stop it is by including an inhibitor in the compound solution ("I" from Fig. 2). Once it starts, corrosion is not easily stopped.
Some inhibitors are effective on ferrous (iron-containing) metals, while others are more effective on nonferrous metals (those not containing iron). Everyone knows that cast iron will rust. So will other ferrous metals, such as powdered iron, hot- and cold-rolled steel, and ductile or hardened steels. Nonferrous metals, such as aluminum, brass, copper, magnesium, zinc and some "stainless" grades do not rust, but may oxidize.
Inhibitors (Fig. 3) are often needed in "threshold" concentrations. Use the manufacturer's recommended minimums. And keep in mind that the active ingredient in an inhibitor is used up as processing continues. So use enough to overcome depletion.
Provide cooling. If a vibratory process were to run dry, friction would generate high temperatures quickly. The solution cools the mass of media and parts. Low flow rates do not carry away heat as fast. Then more oxidation takes place. Elevated temperatures are seldom needed, even for severe cleaning problems.
Be environmentally compatible. Today all solutions should be made of "biodegradeable" surfactants those that degrade so that their foam generating properties are soon naturally reduced to an acceptable level in a stream. Some solutions must leave the plant within certain pH limits, or with a maximum oil content, or a maximum turbidity or metal content. Breaking the emulsion may be required and the oil may haved to be skimmed. The pH is controlled easily by automatic titration. And heavy metals or the solids that cause turbidity can be reduced by pH adjustment, oxidation, reduction, flocculation and other chemical treatments.
Compound Solution and Media
The solution flow rate does control cut rate. This relationship won't go away. It's a fact.
Compound type has an effect on the shape of the curve in Fig. 4. Compounds with high lubricity flatten out the curve, lowering the center of it. High-foam products do the same thing. Some compounds chemically attack the media binder and exaggerate media wear.
"Deburring" compounds are designed to promote cut in vibratory equipment. They do it by control of the following:
- Cleanliness. They must make good cleaning solutions, to keep the media clean. When dirt is allowed, sludge forms and cut rate is reduced. Parts get darker.
- Foam control. It's essential. Too much foam slows down the media action. Too little and the solution won't be held in place to provide its functions.
- Corrosion. Corrosion must be eliminated. The inhibitor need not be too complex, but its benefits far outweigh the minor cost.
- Lubricity. Not too much, or cut stops completely.
Deburring compounds do not contain abrasives to promote cut. Abrasives greatly accelerate media breakdown and are not efficient.
The most important thing to keep in mind about the media-solution relationship is that when it does get out of balance, the problems it causes occur very slowly. This gives a false sense of security to those who do such things as turning down the compound flow rate a touch "to reduce waste." The long times involved often make it difficult for the tinkerer to remember what he did.
Compound Solution and Equipment
The solution must not affect the equipment. It must not corrode it, clog it or otherwise interfere with the function of the machinery. If acid descaling compounds are used, special corrosion-resistant equipment is required. Not just any old machine will do.
Types of compound feed. Three major types of solution-feed systems are important: batch, recirculation, and flow-through.
Batch. This is the simplest system. It is used in barrels and in vibrators with no drains. One simply charges compound and water, runs the process, and then flushes. Other compounds or the same one can be recharged as desired.
Recirculation. Mix a solution in the tank and pump it into the process, allowing it to drain back into the tank for reuse. Sounds efficient, but the high rate of soil production in vibratory finishing creates severe contamination problems. From the first to the last time the solution recirculated through the machine, it was never the same twice. It got dirtier each time through. The active materials were gradually consumed. Later in the cycle more soil is deposited than removed. Compound circulation will never provide steady-state conditions.
Flow-through. This system pumps fresh solution into the machine, allows it to act, then drains it out.
Flow-through solutions generally contain 0.5 to one oz/gal (0.4-0.8 pct or 4-8 ml/liter). They are very dilute and very economical. At 1 ½ gal per cu ft per hr flow rate, a 10-cu-ft machine uses 15 gph. In this water would be 7 ½-15 oz of compound concentrate. It would take from 8 ½ to 17 hr to use a gal of concentrate (2-4 ½ hr per liter).
Compound flow-through means the parts see only clean media. The solution is always the same strength. Parts are clean, too. There is no sludge tank to clean. Flow-through reduces grief and saves money in the long run.
Methods of adding compound. Three methods of compound addition are popular: manual, batch premix and automatic.
With the manual procedure, you put the compound and water in by hand using a measuring cup or other calibrated container.
In the batch premix method the solution is held in a tank where it is mixed prior to being pumped into the machine. The tank can be charged by hand, or this can be done automatically if a compound-metering pump and float-level switches are available.
Automatic addition employs a compound-metering pump and rotameter for measuring flow rate of water. It is the cheapest system, uses the least space (no tank) and requires the least maintenance (fewer parts). Because it works so well, it is recommended.
Largely because the automatic metering system works so well, liquid compounds are used in ever-increasing volumes. Liquids allow a great variety of raw materials to be used and they are extremely economical as a result.
The Compound Solution and the Parts
In Fig. I we saw that the link between the compound solution and the parts says "cleanliness, color, corrosion, speed."
Cleanliness. The surfactants and detergents formulated into compound solutions remove organic residues loosened by the scrubbing action of the media. Emulsification of oils is a very selective process. Cleaning is therefore highly selective as well. There are a great many oily soils. It takes an almost like number of cleaners to remove them efficiently.
If your parts are not clean emerging from this process, first check flow rates and concentration. The best cleaner in the world will do nothing at too low a concentration Fig. 3 shows why threshold concentrations must be maintained. Second, if cleaning still is not sufficient, try other cleaning compounds. It may be necessary to give your supplier a sample of your oils for laboratory screening. Give him a water sample at the same time. These samples will save a lot of trial and error in your plant.
Factors affecting cleaning include type and quantity of soils; compound type; compound concentration; temperature; solution contamination; agitation; time; and rinsing.
Because most of these points have already been covered elsewhere, we need only review them.
Type and quantity of soil. Obvious. Or, it should be by now. Do not allow into your process soil you don't want to pay to remove! Excess soils cost money. If you know what soil is to be removed, the best cleaner formula can be selected and you can use it at lower concentrations.
Compound type and concentration. As noted, selectivity is real. The threshold is different for different products.
Temperature. Higher temperatures help reduce the viscosity of heavy oils and greases. The outstanding scrubbing available in vibratory processes usually offsets need for heating, however, and these processes therefore save energy!
Contamination. Promoted with recirculation; eliminated with flowthrough systems.
Time. Usually very short, on the order of ten min. Any longer and soil is driven back into the parts.
Rinsing. A subject in itself. But let's be sure of one thing: never rinse in plain water! Use an inhibitor! Fig. 5 shows a series of cross-sections through the metal surface and liquid film above it. In 5A, the relatively thin film of compound solution is shown as the part leaves the process. The dots represent the amount of inhibitor in the solution.
Fig. 5B depicts the film after a "good" water rinse. It's a thick film of water containing essentially no inhibitor. Corrosion must start if it can! In 5C we see what happens after inhibiting this water-rinsed part. The inhibitor cannot easily penetrate this thick film of water in the normal time allowed. Water merely dilutes the inhibitor concentration. A relatively thick, poorly inhibited film remains. Corrosion can, and often does, continue.
In 5D we observe what happened to the parts that were dipped into inhibitor without the prior water rinse. The inhibited compound solution is replaced and invigorated by the inhibitor solution. Corrosion can't occur. The inhibitor solution, being a much better wetting solution than the water, actually removes much more surface soil from the surface, and ... cleaner parts!
And now, back to the solution and parts.
Color. The term "color" has two meanings in mass finishing. It can mean the hue or tint of the part, or it can refer to the reflectivity or luster of the part. The solution controls both. And both are the result of the speed of cleaning and the degree of cleaning. Slow cleaners and poor cleaners give darker parts. Soils not removed are beaten back into the surface. Light metal colors are achieved when the solution cleans the surface quickly and keeps it clean during the process.
Corrosion. If surface quality diminishes with time, corrosion is the probable reason. Corrosion will occur on any metal surface not properly inhibited. Corrosion is not easy to detect and it causes problems not normally associated with it: minor defects after plating or painting, adhesion problems, apparent surface roughness in selected areas.
Speed. The solution helps to control cut rate just as machine settings do! Cleaning influences burnish quality, speed, and lightness of color. If the media is clean, the parts will be clean. Under these conditions, speed is maintained.
The Compound Solution and People
One of these days, most mass finishing operators and their supervisors will develop supreme confidence in the consistency of mass finishing. When this happens, most processing problems will disappear. Instead of fighting the problems that occur, operators will realize that the process is quite stupid. It cannot now nor will it ever be able to make a decision that could affect the process! It's just too darned dumb!
People make changes. People change the compound, the media, the water, the oils or mold release, the heat-treat atmosphere, the plating tank, the sharpness of the dies. People adjust solution flow rates, change vibrator speed, or otherwise get the entire thing out of whack. Only people are smart enough to do that. The stupid process can't.
With this recognition comes confidence. With confidence comes the ability to find the reasons for the problems at hand. And with the solutions of problems comes reduced costs, greater productivity, better quality and greater profits. Things people really want!
RELATED CONTENT
-
Fixing Corrosion Between Anodized Aluminum and Steel
Anne Deacon Juhl, Ph.D., with AluConsult, says Galvanic corrosion is due to an electrical contact with a more noble metal or a nonmetallic conductor in a conductive environment.
-
Coating Systems with the Best Long-Term Performance
The best protection against corrosion and UV exposure, says Axalta’s Mike Withers, is electrocoat and a super durable powder coating.
-
Pretreatment for Painting
Better adhesion, enhanced corrosion and blister resistance, and reduced coating-part interactions make pretreatment a must.