UV Curing
A UV curing process can provide finishers a number of advantages, including reduced floorspace required, increased productivity and a high-quality finish . . .
Ultraviolet curing-the process of transforming a liquid into a solid by the use of light-is an energy efficient and relatively low-temperature technology with many applications in coating, printing, adhesives, electronics and communication products. UV curing provides improved overall physical or chemical properties of polymeric materials and produces superior results in bonding, surface finish and durability. It is used on virtually all substrates, including plastics, paper, film and foil, wood, metal, glass, fiber and composite. Speed and controllability in a variety of applications are driving increasing worldwide markets for this proven technology.
Benefits
The UV curing market is growing rapidly because it is a "clean" technology that increases productivity compared to other traditional methods of curing. The UV curing process has a number of key attributes:
Featured Content
- no solvents-because cure is by polymerization rather than by evaporation, VOC and HAP emissions are virtually eliminated;
- low temperature-heat is not required;
- high speed-cure is nearly instantaneous;
- energy efficient-energy is invested only in the curing reaction, not in heating;
- easily controlled-since inks and coatings do not "dry," they don't set up in printing/coating equipment or change viscosity; and
- quality finishes-superior resistance to scratches and chemicals.
Applications
UV processing offers several advantages over other finishing methods. Typical product lines involve coatings (on wood, metal, paper and plastic), inks (for letterpress, lithographic, gravure and screen printing) and adhesives (for film, foil or paper substrates). The industries using these technologies are diverse and varied, including automotive components, medical products, electronics, CDs and DVDs, two-piece and three-piece can printing, pipe and tube coating, furniture, fiber optics, flooring, packaging and containers.
Thermal vs. UV Curing
Solvent-borne, thermally cured inks or coatings are composed of a resinous binder, pigments, fillers and diluent solvents. After application to the substrate, heat is applied to drive off the solvents and dry the coating film. The evaporated solvents are generally flammable and toxic and become airborne pollutants. Solvent emissions often require the use of more energy and capital investment to be incinerated or scrubbed and recovered by distillation.
In contrast, the UV curing process achieves the transition from liquid to solid by means of chain-addition polymerization. This polymerization is triggered by the reaction of a low concentration of an ingredient called a photoinitiator, which absorbs and reacts to UV light. In UV-curable coatings, the resin binder is replaced by a formulation of liquid monomers and oligomers, which are induced to crosslink by the reaction of the photoinitiator. To create an ink, pigments may be dispersed in the liquid formulation. The coating is completely reactive and the thickness that is laid down wet is essentially the same as the thickness after curing. In UV inks, the pigment does not enter into the crosslinking reaction but is literally locked in place. There is no mass transfer and no evaporation of solvents-just wet to dry by exposure to a UV light source.
Technology of UV Curing
Typically, the radiant energy produced by the UV lamp is focused by a reflector onto the coating or ink. The UV energy striking the surface causes the photoinitiator to trigger the polymerization reaction. The material is usually solidified or dried when it exits the UV cure zone. The time, and consequently the space, required for cure is significantly less than thermal drying methods. Because the process relies on UV light to initiate the crosslinking of molecules, it does not evaporate any solvents nor significantly heat the substrate.
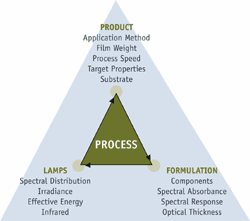
Key Components
A UV Curing system consists of three component parts all integrally related. The end product determines the requirements of the physical properties of the cured photochemistry. Target properties, such as opacity or hiding, film thickness, hardness or flexibility, chemical resistance, resistance to abrasion or scratching and adhesion, are only a few of the properties that may represent the performance requirements of the application.
The photochemistry is designed to achieve the target properties upon exposure to the appropriate UV energy. Formulation variables include a combination of monomers, oligomers, photoinitiators and functional additives. The optical characteristics of the formulated material determine the most effective lamps to use.
The UV lamp system has a number of variables, such as power, wavelength and focus, that will also have a significant affect on the target properties of the end product. The selection of these variables depends on the optical properties of the formulated material.
These three components -application method, formulation and cure exposure-are coordinated to create a successful UV cured system.
Categories of UV Curing
A variety of materials and product handling methods are involved in UV curing, so the part size, surface area and speed of the process will require curing systems designed to accommodate them. UV curing can be divided into several categories distinguished by the size of the work surface and the type of motion of the work piece.
Linear Curing. Linear processing is the most common arrangement for curing and is typically used to cure flat surfaces. The coated surface is passed under or by UV lamps to expose the surface. Printing presses, roll-to-roll coaters and conveyors are all variations on linear processing. Typically, a tubular lamp with a focusing reflector or rows of tubular lamps extend across the surface, providing uniformity of UV exposure in that dimension, while the motion of the work surface provides uniformity of exposure along the direction of travel. Because the lamps can be arranged closely to the surface, a very high intensity of UV light can be achieved.
Flood Curing. The simplest method of UV exposure is to place an object or surface under a UV lamp and control the time during which it is exposed. This is often referred to as static curing. It is frequently used for laboratory exposure or for low-volume production curing. Flood exposure is regularly used in film transfer, printing plate making and printed circuit manufacturing.
However, when the surface is complex or curved, or even 3D, it becomes more difficult to cover the surface in a linear fashion with focused high intensity light. The illumination may be lower in intensity as the energy is distributed over a larger area. By combining additional degrees of motion of the part, such as rotating while passing through the curing lamp region, a complex surface may be easily and adequately cured. Further, lamps of various configurations can be used, depending on type and degree of motion, size and complexity of the surface contours.
Spot Curing. A small, high intensity spot of UV light directed precisely at a work point characterizes spot curing. It is typically used for UV bonding of adhesives for medical product assembly and electronic assembly. The light is transmitted to the work area by means of a liquid light guide with a projection lens at the tip. This enables the spot cure system to work in spaces and with small assemblies where a large lamp would be impractical.
Cost Considerations
The use of UV curing presents a number of economic benefits over alternative methods. An economic benefit analysis for any one application may be substantially different from another. While they will vary with each application, cost savings may result from such factors as higher mileage yield from 100% solids inks and coatings, reduced setup time, reduced waste and scrap, better use of space, reduction of work-in-process, increased productivity, energy savings, increased yield, reduction or elimination of VOC processing and improved product quality and performance.
Where are We?
UV curing is a proven process, used in a large variety of industries. It has displaced or replaced coating methods using conventional solvent-borne systems because of reduced pollution, lower cost, higher product quality, higher production speed and lower space consumption. Many of these have become possible because of the development of new commercially available UV-curable chemistry systems.