Can Electroplated Fe-C be an Environmentally Friendly Alternative to Hard Chromium and DLC Coatings?
Electroplated FeC is an efficient surface treatment based on non-aggressive chemicals with a deposition rate of ~20 μm/h at a process temperature of 50°C. The FeC coating is carbide-free and temperature stable up to ~250°C with a hardness of 750 HV, which is comparable to frequently applied hardened steels. The FeC coating has reasonable friction properties and have high affinity towards lubricants because of incorporated amorphous carbon. Hence, for certain applications, the FeC coating might be an interesting wear-protective alternative to hard chromium and to PVD-deposited low-friction diamond-like carbon coatings (DLC’s), which are rather difficult and costly to deposit on larger items.
#research #surfin #vacuum-vapor
A Synopsis* of a Presentation given at SUR/FIN 2017 (Atlanta, Georgia)
by
Featured Content
Lars Pleth Nielsen**
Tribology Center,
Danish Technological Institute, Aarhus, Denmark
&
Jacob Obitsø Nielsen and Per Møller
Technical University of Denmark, Lyngby, Denmark
Editor’s Note: The following is a synopsis of a presentation given at NASF SUR/FIN 2017, in Atlanta, Georgia on June 19, 2017 in Session 2, Advances in Surface Finishing Technology I. This brief can be accessed and printed HERE; the complete Powerpoint presentation is available by clicking HERE.
ABSTRACT
Electroplated FeC is an efficient surface treatment based on non-aggressive chemicals with a deposition rate of ~20 μm/h at a process temperature of 50°C. The FeC coating is carbide-free and temperature stable up to ~250°C with a hardness of 750 HV, which is comparable to frequently applied hardened steels. The FeC coating has reasonable friction properties and a presumed high affinity towards lubricants because of incorporated amorphous carbon. Hence, for certain applications, the FeC coating might be an interesting wear-protective alternative to hard chromium and to PVD-deposited low-friction diamond-like carbon coatings (DLC’s), which are rather difficult and costly to deposit on larger items. The mechanical properties, temperature stability, friction and corrosion properties of electroplated FeC are compared with PVD deposited a-C:H and a newly developed a-C:H:Si DLC coating. The comparisons are based on scratch tests, friction measurements, SEM, TEM, Raman and corrosion studies.
Hard chromium
Dr. Nielsen first discussed the nature of hard chromium and its importance to commerce. Its many useful properties were deemed difficult to replace in one single solution. Among those cited were its high hardness, its ability to deposit large thicknesses up to 100 μm, a low process operating temperature compared to PVD and thermochemical diffusion processes, its corrosion resistance, its surface lubricity, as well as easy release properties in plated molds via low surface energy.
As a result of chemical regulatory protocols, REACH in particular, hexavalent chromium requires authorization since it is listed on the REACH Annex XIV. The sunset date for using chromium trioxide (Cr+6) in Europe was September 21, 2017. It is no longer allowed to use hexavalent chromium for any production in the surface treatment field without having an authorization.
Given this, Dr. Nielsen discussed the use of electroplated Fe-C as an alternative. The FeC coating had reasonable friction properties and has a high affinity towards lubricants because of incorporated amorphous carbon. Hence, for certain applications, the FeC coating might be an interesting wear-protective alternative to hard chromium and to PVD-deposited low-friction diamond-like carbon coatings (DLC’s), which are rather difficult and costly to deposit on larger items. In this presentation, he reviewed studies of three possible alternatives, including electroplated Fe-C; a diamond-like carbon (DLC) coating doped with hydrogen – a-C:H – by PVD; and a PVD DLC a-C:H coating with the addition of silicon – a-C:H:Si.
Electroplated Fe-C and its properties
The Fe-C electrolyte was based on a solution of ferrous sulfate containing a stabilizing agent. An inert titanium-platinum anode was used. The operating temperature range was 30 to 60°C (nominally 50°C), and the operating current efficiency was ~80%. Bath operation was quite stable, allowing continuous electrodeposition from a 25-L bath for 24 hr at a stable pH with no chemical adjustments. The deposition rate was 23 μm/hr.
Using glow-discharge optical emission spectroscopy (GDOES), the average carbon content was 0.85 wt% (Fig. 1). X-ray diffraction (XRD) showed that the as-deposited coating consisted of ferrite with a minor amount of Fe2O3. One-hour annealing studies showed this structure to be stable up to 300°C, above which (315°C), the structure consisted of ferrite, β-Fe2O3, Cohenite (Fe3C) plus undefined phases (Fig. 2).
The hardness was particularly high, with average the microhardness test result at 750 HV. This value remained stable with annealing up to 300°C (Fig. 3).

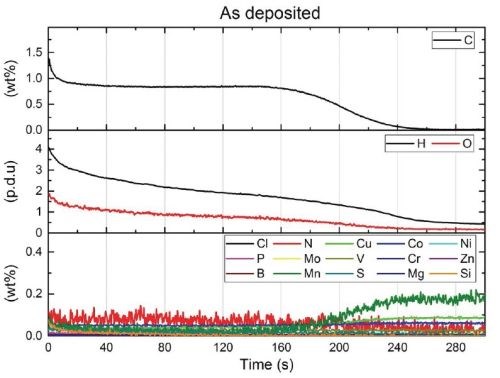
Figure 1 - GDOES results: as deposited FeC electrodeposit.

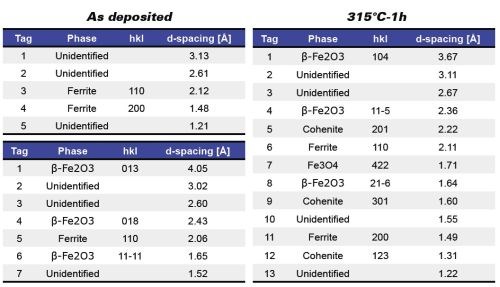
Figure 2 - Fe-C deposit structure, both as-deposited and annealed at 315°C by TEM diffraction.

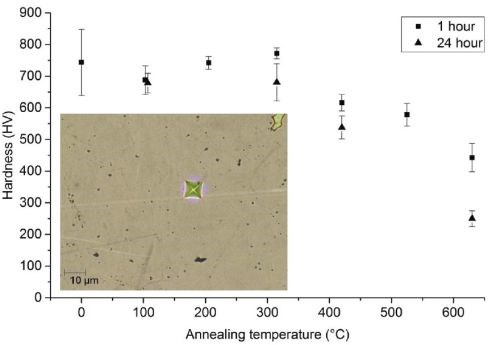
Figure 3 - Fe-C deposit hardness versus annealing temperature (1 and 24 hr).
PVD diamond-like carbon coatings and their properties
The DLC coatings were produced by DC magnetron sputtering in an argon-nitrogen-acetylene atmosphere. A CrN adhesion layer was first deposited after which the a-C:H and a-C:H:Si layers were deposited (Fig. 4). The deposition temperature was about 200°C and the thickness produced, including the adhesion layer, was about 3 μm.
In comparison, the DLC coatings were considerably harder than electroplated Fe-C. The a-C:H coating exhibited a hardness of 1600 HV. This value held at an annealing temperature of 200°C for two hours in air; however, the coating delaminated at 300°C. By contrast, the as-deposited a-C:H:Si coating exhibited a hardness of 2200 HV, which was maintained in the annealing regime up to 300°C. In sum, the DLC coatings were much harder than the Fe-C, but the a-C:H:Si DCL was better than the a-C:H coating in terms of hardness stability during annealing.


Figure 4 - Cross-section of PVD diamond-like carbon coatings: (L) a-C:H, (R) a-C:H:Si; the schematic shows the initial CrN adhesion layer and the transition gradient.
Tribology performance
Dr. Nielsen reported on the tribological studies for the three coatings, with hard chromium samples used as a benchmark. Both the scratch adhesion and pin-on-disk wear tests were used in this phase of work.
The scratch adhesion test used a progressive load along the scratch from 0.10 to 30.0 N. The results are shown in Fig. 5, compared with hard chromium. Both thin and thick deposits of the electroplated Fe-C coating were evaluated. The Fe-C showed adhesive failure, with the thin coating performance visibly worse (Fig. 6). The team is currently improving the adhesion. The a-C:H:Si DLC coating performed better than the a-C:H material (labeled “DLC-TR”).


Figure 5 – Scratch test results.


Figure 6 – Comparison of scratch test results for thick and thin Fe-C coatings.
The pin-on-disk wear tests used a normal load of 5 N, a sliding velocity of 0.637 m/sec and a duration of 4 hr. The loading pins consisted of AISI 420 stainless steel for all samples. In addition Al2O3 pins were used on the Fe-C and a-C:H:Si coatings. The coefficient of friction was determined as the average value in the 2 to 3-hr interval of the 4-hr test.
The FeC coatings delaminated under the test conditions due to adhesion of the FeC to the Al2O3 test ball. The coefficients of friction (AISI 420) for the a-C:H and a-C:H:Si coatings were 0.131 and 0.084, respectively. This compared to 0.890 for hard chromium. The respective wear rates (AISI 420) were 1.781E-07 and 5.585E-07 mm3/Nm (compared with 1.124E-5 for hard chromium.
Summary
The Fe-C electrodeposits exhibited a high hardness of ~750 HV, and were stable on heating up to 300°C. They contained a relatively high content of homogeneously distributed carbon. If the coating was too thin however, it was vulnerable to cracking under pressure. Coating improvement and further tests are in progress.
In pin-on-disk testing, the DLC a-C:H:Si PVD coating exhibited the lowest friction in air. DLC a-C:H showed friction to be dependent on pressure, but it exhibited the lowest wear rate. The Fe-C deposits did exhibit adhesive failures. Interestingly, hard chromium showed high wear.
Taken together:
| Wear | a-C:H < a-C:H:Si << Hard Cr |
| Friction in air | a-C:H:Si < a-C:H << Hard Cr |
| Friction in vacuum | a-C:H < a-C:H:Si << Hard Cr |
| Temperature stability | a-C:H < a-C:H:Si < FeC < Hard Cr |
About the author

.jpg;maxWidth=600)
Footnotes
*Compiled by Dr. James H. Lindsay, NASF Technical Editor
**Presentation author:
Dr. Lars Pleth Nielsen, Director
Tribology Center,
Danish Technological Institute
Kongsvang Alle 29
Aarhus, Aarhus 8000 Denmark
Phone: +45 72201585
Mobile: +45 72201585
Email: lpn@dti.dk
RELATED CONTENT
-
History of Chromium Plating
A review article originally printed as Plating & Surface Finishing, 71 (6), 84-91 (1984) on the occasion of the 75th anniversary of the founding of the American Electroplaters Society.
-
An Improvement in the Zincate Method for Plating on Aluminum
Investigations were conducted into the sodium zincate solution used for treating aluminum prior to its being plated. Measurements of the adhesion of the nickel plate to aluminum and its alloys were done to check the effect of modifications made to this solution.
-
Electroplated Tin-Nickel Coatings as a Replacement for Nickel to Eliminate Nickel Dermatitis
This paper is a peer-reviewed and edited version of a paper delivered at NASF SUR/FIN 2013 in Rosemont, Ill., on June 12, 2013.


















