Electro-codeposition of MCrAlY Coatings for Advanced Gas Turbine Applications - 3rd Quarterly Research Report
This NASF-AESF Foundation research project report covers the third quarter of project work (July-September 2018) on this AESF Foundation Research project at the Tennessee Technological University. The objective of the work is to study and optimize the MCrAlY electro-codeposition process to improve the coating oxidation/corrosion performance. In this quarter, studies continued on the effect of current density on CrAlY particle incorporation in electro-codeposited Ni-CrAlY composite coatings, focusing on the current density range of 20 to 60 mA/cm2.
#research #nasf
3rd Quarterly Report
July-September 2018
Featured Content
AESF Foundation Research Project #R-119
Electro-codeposition of MCrAlY Coatings
for Advanced Gas Turbine Applications
by
Prof. Ying Zhang* & J.C. Witman
Department of Mechanical Engineering
Tennessee Technological University
Cookeville, Tennessee, USA
Editor’s Note: This NASF-AESF Foundation research project report covers the third quarter of project work (July-September 2018) on an AESF Foundation Research project at the Tennessee Technological University, Cookeville, Tennessee. A printable PDF version of this report is available by clicking HERE.
Summary
In this quarter, we continued to study the effect of current density on CrAlY particle incorporation in electro-codeposited Ni-CrAlY composite coatings, focusing on the current density range of 20 to 60 mA/cm2. Both ball-milled and gas-atomized CrAlY powders were used and the Ni-based alloy specimens were plated in a rotating barrel. For ball-milled CrAlY and CrAlYTa powders, the particle incorporation in the as-deposited coatings decreased from ~40 to 25-33 vol% as the current density was increased from 5 to 40 mA/cm2. No further decrease in particle incorporation occurred when the current density was increased further, from 40 to 60 mA/cm2. For gas-atomized CrAlY powder, the current density appeared to have little influence on the particle incorporation, while an overall higher particle volume percentage (40-43 vol%) was observed for the entire current density range (10-60 mA/cm2) employed in this study.
Technical report
I. Introduction
To improve high-temperature oxidation and corrosion resistance of critical superalloy components in gas turbine engines, metallic coatings such as diffusion aluminides or MCrAlY overlays (where M = Ni, Co or Ni+Co) have been employed, which form a protective oxide scale during service.1 The state-of-the-art techniques for depositing MCrAlY coatings include electron beam-physical vapor deposition (EB-PVD) and thermal spray processes.1 Despite the flexibility they permit, these techniques remain line-of-sight which can be a real drawback for depositing coatings on complex-shaped components. Further, high costs are involved with of the EB-PVD process.2 Several alternative methods of making MCrAlY coatings have been reported in the literature, among which electro-codeposition appears to be a more promising coating process.
Electrolytic codeposition (also called “composite electroplating”) is a process in which fine powders dispersed in an electroplating solution are codeposited with the metal onto the cathode (specimen) to form a multiphase composite coating.3,4 The process for fabrication of MCrAlY coatings involves two steps. In the first step, pre-alloyed particles containing elements such as chromium, aluminum and yttrium are codeposited with the metal matrix of nickel, cobalt or (Ni,Co) to form a (Ni,Co)-CrAlY composite coating. In the second step, a diffusion heat treatment is applied to convert the composite coating to the desired MCrAlY coating microstructure with multiple phases of β-NiAl, γ-Ni, etc.5
Compared to conventional electroplating, electro-codeposition is a more complicated process because of the particle involvement in metal deposition. It is generally believed that five consecutive steps are engaged:3,4 (i) formation of charged particles due to ions and surfactants adsorbed on particle surface, (ii) physical transport of particles through a convection layer, (iii) diffusion through a hydrodynamic boundary layer, (iv) migration through an electrical double layer and finally, (v) adsorption at the cathode where the particles are entrapped within the metal deposit. The quality of the electro-codeposited coatings depends upon many interrelated parameters, including the type of electrolyte, current density, pH, concentration of particles in the plating solution (particle loading), particle characteristics (composition, surface charge, shape, size), hydrodynamics inside the electroplating cell, cathode (specimen) position, and post-deposition heat treatment if necessary.3-6
There are several factors that can significantly affect the oxidation and corrosion performance of the electrodeposited MCrAlY coatings, including: (i) the volume percentage of the CrAlY powder in the as-deposited composite coating, (ii) the CrAlY particle size/distribution, and (iii) the sulfur level introduced into the coating from the electroplating solution. This three-year project aims to optimize the electro-codeposition process for improved oxidation/corrosion performance of the MCrAlY coatings. The three main tasks are as follows:
- Task 1 (Year 1): Effects of current density and particle loading on CrAlY particle incorporation.
- Task 2 (Year 2): Effect of CrAlY particle size on CrAlY particle incorporation.
- Task 3 (Year 3): Effect of electroplating solution on the coating sulfur level
In this reporting period, we continued to study the effect of current density on CrAlY particle incorporation in electro-codeposited Ni-CrAlY composite coatings, focusing on the current density range of 20 to 60 mA/cm2.
II. Experimental Procedure
2.1. Substrate alloys and powders
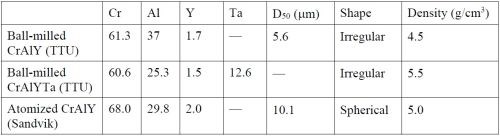
Substrates were made from available nickel-based alloys including Ni 200 (>99.0 Ni, with 0.25 Cu-0.40 Fe-0.35 Mn-0.15 C-0.35 Si-0.01 S max., in wt%) and René 80 (Ni-3.0 Al-14.1 Cr-9.7 Co-4.3 W-4.0 Mo-5.0 Ti-0.18 C in wt%, 130B-200 Zr-7 S in ppmw). Disc specimens (1.6 mm thick, ~17 mm in diameter) were cut with an abrasive cutting saw. The specimens were ground to #600 grit using SiC grinding papers, followed by grit blasting with #220 Al2O3 grit, and were then ultrasonically cleaned in hot water and acetone. Pre-alloyed CrAlY-based powders (Table 1) were used in the electro-codeposition experiments, including the laboratory ball-milled CrAlY and CrAlYTa powders made at TTU, and the commercial gas-atomized CrAlY powder purchased from Sandvik.
Table 1 - Compositions (wt%) and properties of the powders used in the electro-codeposition experiments.
2.2. Electro-codeposition
A rotating barrel system (Fig. 1) was employed in the electro-codeposition experiments; details can be found in the 2018-Q1 Report. Watts nickel plating solution was used, and the nickel anode was placed outside of the barrel along with a mechanical stirrer and heating coil. The specimens were plated at 50°C with a pH level of 3.7-3.9. The CrAlY particle concentration in the plating bath was kept at 20 g/L and the barrel rotation speed at 7 RPM. To understand the effect of current density on particle incorporation, specimens were plated at different current densities, ranging from 5 to 60 mA/cm2. The plating time was varied to ensure that similar coating thicknesses were obtained.
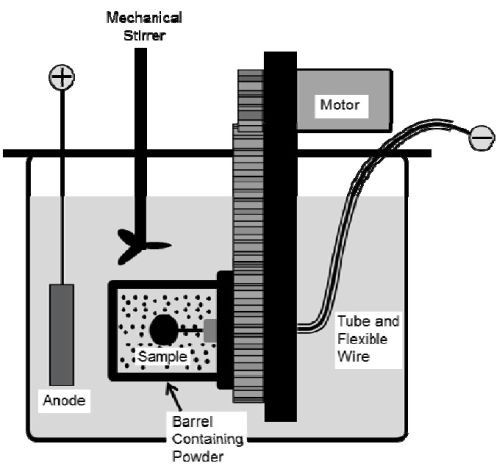
Figure 1 - Schematic of the barrel system.
2.3. Coating characterization
The Ni-CrAlY(Ta) composite coatings were characterized using scanning electron microscopy (SEM) equipped with energy dispersive spectroscopy. Prior to metallographic sample preparation, the specimens were copper-plated to improve the edge retention. To determine the volume fraction of the incorporated CrAlY particles, multiple backscattered electron images were taken from different locations along the coating cross-section, which were then processed using the ImageJ software. The brightness and contrast of the image were adjusted by setting a proper threshold such that the particles were separated from the background. The area fraction of the CrAlY particles was determined, which was assumed equivalent to its volume fraction.
III. Results and discussion
Typical MCrAlY coatings contain 8-12% Al, 18-22% Cr, and up to 0.5% Y (in wt%). To form a NiCrAlY coating with 10 wt% Al, based on the chemical composition and the density of the CrAlY powder used in this study, approximately 40 vol% of CrAlY particles were needed in the as-deposited composite coatings.6 In addition to achieving a uniform and adherent coating, incorporation of a high percentage of CrAlY particles in the nickel matrix is another important requirement for the electro-codeposition process.
Figure 2 shows the cross-sections of Ni-CrAlY composite coatings deposited with atomized CrAlY powders at two different current density levels, i.e., 10 and 60 mA/cm2. The coating thickness was in the range of 100-120 μm. The spherical shape of the gas-atomized powders can be clearly seen, which contrasted with the irregular geometries of the ball-milled powders, as presented in the 2018-Q2 Report.

Figure 2 - SEM cross-sectional images of the Ni-CrAlY coatings fabricated with the atomized CrAlY powders at different current densities: (a) 10 mA/cm2; (b) 60 mA/cm2.
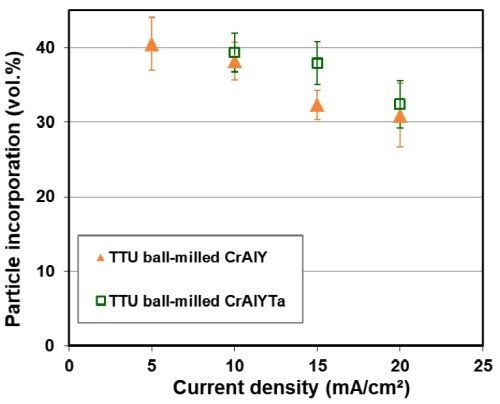
The effect of current density on CrAlY(Ta) particle incorporation is displayed in Fig. 3. For ball-milled CrAlY and CrAlYTa powders, the particle incorporation in the as-deposited coatings decreased from ~40 to 30-33 vol% as the current density was increased from 5 to 20 mA/cm2 (Fig. 3a). The particle incorporation continued to drop to 25 vol% when the current density was further increased to 40 mA/cm2 and remained at ~25 vol% for current densities between 40 and 60 mA/cm2 (Fig. 3b). For gas-atomized CrAlY powder, the current density appeared to have little influence on the particle incorporation, while an overall higher particle volume percentage (40-43 vol%) was observed for the entire current density range (10-60 mA/cm2) employed in this study.
(a)
(b)
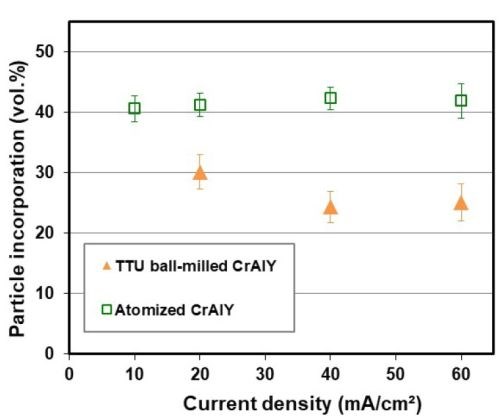
Figure 3 - Particle incorporation in the coating as a function of plating current density: (a) in the low current density range with ball-milled CrAlY and CrAlYTa powders, and (b) in the high current density range with ball-milled and atomized CrAlY powders.
Current density is a critical process parameter in conventional electroplating that governs the deposition rate. However, the effect of current density on particle incorporation depends strongly on the nature of particles and the metal deposit. Different types of relationships have been observed,7 i.e., the particle content in the composite coatings either increases or decreases continuously with the current density or exhibits one or multiple peaks as a function of current density.
Our present findings for gas-atomized CrAlY powder are in good agreement with an earlier study conducted by Foster, et al.8 for coatings plated under several conditions using CrAlY particles in a rotating barrel configuration. Similar results were also observed by Liu and Chen for Ni-Al composite coatings made with sediment codeposition,9 in which the current density showed minimal influence on the aluminum particle incorporation between 20 and 90 mA/cm2. The current density dependence for conducting particles is generally different than non-conducting particle systems such as Ni-Al2O3 and Ni-SiC, as reported in the literature.10,11
Current density also has an effect on the overall coating quality by affecting the porosity and thickness uniformity. Higher current densities typically result in preferential deposition at the edges and corners of the specimen, which can cause higher stresses and consequently coating cracking/spallation in those areas. On the other hand, at lower current densities, if the rate of metal deposition cannot keep up with the speed of particle settlement, there is a tendency to form less dense coatings with noticeable voids.
IV. Future work
In the next quarter, the concentrations of CrAlY powders in the plating solution will be varied to study the effect of particle loading on particle incorporation in the electro-codeposited coatings.
References
1. G.W. Goward, Surf. Coat. Technol., 108-109, 73-79 (1998).
2. A. Feuerstein, et al., J. Therm. Spray Technol., 17 (2), 199-213 (2008).
3. C.T.J. Low, R.G.A. Wills and F.C. Walsh, Surf. Coat. Technol., 201 (1-2), 371-383 (2006).
4. F.C. Walsh and C. Ponce de Leon, Trans. Inst. Metal Fin., 92 (2), 83-98 (2014).
5. Y. Zhang, JOM, 67 (11), 2599-2607 (2015).
6. B.L. Bates, J.C. Witman and Y. Zhang, Mater. Manuf. Process, 31 (9), 1232-1237 (2016).
7. A. Hovestad, R.J.C.H.L. Heesen and L.J.J. Janssen, J. Appl. Electrochem., 29 (3), 331-338 (1999).
8. J. Foster, B.P. Cameron and J.A. Carew, Trans. Inst. Met. Finish., 63 (3-4), 115-119 (1985).
9. H. Liu and W. Chen, Surf. Coat. Technol., 191 (2-3), 341-350 (2005).
10. K. Barmak, et al., J. Microsc., 185 (2), 265-274 (1997).
11. S.C. Wang and W.C.J. Wei, Mater. Chem. Phys., 78 (3), 574-580 (2003).
Past project reports
1. Quarter 1 (January-March 2018): Summary: NASF Report in Products Finishing; NASF Surface Technology White Papers, 82 (12), 13 (September 2018); Full paper: http://short.pfonline.com/NASF18Sep1.
2. Quarter 2 (April-June 2018): Summary: NASF Report in Products Finishing; NASF Surface Technology White Papers, 83 (1), 13 (October 2018); Full paper: http://short.pfonline.com/NASF18Oct1.
About the author

Dr. Ying Zhang is Professor of Mechanical Engineering at Tennessee Technological University, in Cookeville, Tennessee. She holds a B.S. in Physical Metallurgy from Yanshan University (China)(1990), an M.S. in Materials Science and Engineering from Shanghai University (China)(1993) and a Ph.D. in Materials Science and Engineering from the University of Tennessee (Knoxville)(1998). Her research interests are related to high-temperature protective coatings for gas turbine engine applications; materials synthesis via chemical vapor deposition, pack cementation and electrodeposition, and high-temperature oxidation and corrosion. She is the author of numerous papers in materials science and has mentored several Graduate and Post-Graduate scholars.
Jason C. Whitman is a Ph.D. student in the Department of Mechanical Engineering, Tennessee Technological University, in Cookeville, Tennessee. His primary work is involved with the AESF Foundation Research Project R-119, Electro-codeposition of MCrAlY Coatings for Advanced Gas Turbine Applications.
*Corresponding author:
Dr. Ying Zhang, Professor
Department of Mechanical Engineering
Tennessee Technological University
Cookeville, TN 38505-0001
Tel: (931) 372-3265
Fax: (931) 372-6340
Email: yzhang@tntech.edu
RELATED CONTENT
-
Mechanical Properties of Electroformed Metals
In 1996, the AESF held its highly regarded electroforming course, prepared by Ron Parkinson for presentation by The Nickel Development Institute (NiDI) and the AESF. What follows is a slightly modified excerpt, specifically on the mechanical properties of electroformed metals. Much of this information has withstood the test of time, and gives a perspective of this technology at the turn-of-the-century.
-
The Study of Copper Anodes in Acid and Cyanide Plating Baths
The 1956 Carl E. Huessner Gold Medal Award was given to Charles Faust and William H Safranek for Best Paper appearing in Plating or the AES Technical Proceedings in 1955, and their paper is republished here in a series on the AES/AESF/NASF Best Paper Awards. Their work involves an evaluation of anodes for copper plating at the time when OFHC anodes were first emerging in use.
-
Hybrid Sol-Gel Coatings in Surface Engineering
A look at the use of modified sol-gel polymer films and hybrid system coatings, as well as the methodologies for evaluating the mechanical properties of the coatings.


















