Electroless Nickel-Plated Steel vs. Stainless Steel
Corrosion resistance comparisons and cost savings of EN-plated mild steel versus 400 series stainless steel.
Corrosion resistant steels have been in existence for well over a thousand years. One of the earliest examples is the Iron Pillar of Delhi, a Hindu monument constructed around A.D. 400 from an iron-, and interestingly enough, phosphorus alloy. Its longevity and corrosion resistance are due to the formation of a phosphate film. After 1,600 years in the open air, it has barely corroded, and the film has grown by just 1/20 of a millimeter.
There are many other instances of historic corrosion resistant steels, but commercial, large production practicality has come only in the last 100 years with the development of the electric arc furnace, in which most of today’s stainless steels are produced.
Featured Content
Most stainless steels today owe their stain and corrosion resistance to high levels of chromium and nickel in the alloy. There are more than 150 grades of stainless steel available on the market, classified in five groups: austenitic, martensitic, ferritic, precipitation-hardened and duplex. This study only considers the austenitic alloys 303 and 304, and the martensitic alloy 416R.
Austenitic stainless steels (200 and 300 series) are generally more corrosion resistant than the martensitic stainless alloys, but as a rule, cannot be hardened by heat treatment. Martensitic alloys (400 series) can be hardened by heat treatment, but usually are not as corrosion resistant as the austenitic alloys. Composition comparisons are shown in Table 1.
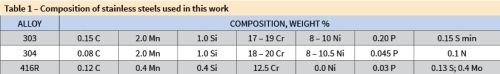
Table 1
Drawbacks to using stainless steels in manufacturing include price, machinability and hardenability. According to worldsteelprices.com, during April of 2014, the Global Composite Carbon Steel price was $713/ton, where the Global Composite Stainless Steel 304 price was $2,778/ton. Stainless steel costs upwards of 3.8 times as much as carbon steel.
In terms of machinability, as a class, stainless steels are generally more difficult to machine than carbon or low-alloy steels because of their higher strength and higher work hardening rates. They require greater power and lower machine speed, while shortening tool life and sometimes leading to difficulty in obtaining a fine surface finish. However, wide variations exist in these characteristics among the different types of stainless steels.
Concerning hardenability, as previously stated, austenitic stainless steels generally cannot be hardened by heat treatment. Because of these drawbacks, at least two manufacturers have either switched, or considered switching to, electroless nickel (EN)-plated mild steel. What follows are two case studies related to these manufacturers’ experience.

A major firearms manufacturer producing sporting rifles with stainless steel barrels and bolts also considered cost reduction strategies to maintain or increase its market position while still supplying customers with outstanding value.
Case Study 1: Manufacturer of Hydraulic Load-Leveling Systems
Iowa-based HWH Corp. is a manufacturer of load-leveling systems for recreational vehicles and campers, among other things. The company was founded in 1967 and for the first 17 years of business, it designed and manufactured machines and hydraulics for agricultural customers, including John Deere and Caterpillar.
During those early years in the agricultural industry, it also discovered a niche for supplying hydraulic leveling systems to the RV industry and has since led in that technology. It has introduced countless novel hydraulic slide-out systems and hydraulic/air solutions to the RV market, ranging from hydraulic step systems to air suspension and saddle-rack systems.
Most of the hydraulic shafts were made from 303 and 304 stainless steel, and while performing well, they were expensive to manufacture. Further, during the early and mid-2000s, stainless steel prices rose sharply, and availability decreased dramatically. HWH began to look for alternatives, and EN was one of them.
After many discussions with management and engineering staff at HWH, it was decided that 1026 and 1018 steel tubing plated with 0.0015-inch thick high-phosphorus EN would be evaluated, with parts baked for stress and hydrogen embrittlement relief. Some of these parts would be put into service right alongside the stainless steel shafts to see if there were any performance differences; others would be left outside for extended periods to gauge the relative corrosion protection characteristics.
After months of successful testing, it was decided to replace the stainless with EN-plated 1026 mild steel on one series of the hydraulic load leveling jacks. A small EN line was installed and, after overcoming the plating learning curve, limited production began.
After one year with a zero failure rate, the decision was made to install a large line and change all the stainless jacks to EN-plated 1026 and 1018 steel—a process that took an additional year. HWH now plates all its hydraulic jack shafts with 0.0015-inch high phosphorus EN. It uses the bath to about five metal turnovers, at which point the older bath is used to EN plate other system parts that would not otherwise get plated, providing additional customer value. The company coats hundreds of shafts and hundreds of other parts each day.
During the first full year of change from stainless to EN plated steel, the company calculated savings of over $1 million.


Figure 3: Specimens under study in the as-received condition: on the left, magazine boxes with stainless on the left and EN on the right; in the right photo, barrels with EN on top and stainless on the bottom.
Case Study 2: Firearms Manufacturer
A major firearms manufacturer producing sporting rifles with stainless steel barrels and bolts also considered cost reduction strategies to maintain or increase its market position while still supplying customers with outstanding value. It investigated the use of EN-plated steel in place of the 416R stainless alloy it was using on some of its extreme environment rifles. This manufacturer already had experience with the use of EN to coat gun parts for aesthetics, corrosion resistance, lubricity and wear. Satisfied it would meet those criteria, the company needed to determine if, for these specific parts, EN would meet or exceed the corrosion performance of the 416R series stainless barrel and bolt, and the 304 series magazine.
One rifle barrel, bolt and bolt plug were fabricated from 1155 steel and plated with a duplex EN coating of 0.0003 inch of mid-phosphorus EN under 0.0002 inch of high phosphorus EN, for a total thickness of 0.0005 inch. The components were assembled into a working gun with stock and fire control mechanism. Each of the three different style magazine boxes were fabricated from 1010 steel and coated with 0.0002–0.0004 inch of high phosphorus EN. The magazine boxes were not assembled into guns.
These boxes were placed in a salt spray cabinet along with a 416R stainless steel extreme-condition gun and the corresponding 304 stainless magazine boxes. Photos were taken, and the guns were checked for functionality at 4, 45 and 93 hours of salt spray exposure. The results follow:
Specimens in As-Received Condition: Figure 3 shows the gun parts in the as-received condition, labeled as stainless steel or EN-plated. The yellowish cast taken on by the EN finishes is merely a reflection of the surrounding area in the laboratory.
Specimens after 4 hours of salt spray: Figure 4 shows the test parts after four hours of salt spray, labeled as stainless steel or EN-plated. The first signs of corrosion are already apparent on the stainless steel barrel. Mechanically, both actions were fully functioning. No corrosion was noted on the EN-plated receivers or bolts. Magazine box A showed no rust on the stainless steel finish. Very slight rusting was noted on the shelf area of the EN-plated box. Boxes B and C exhibited no corrosion.


Figure 4: Corrosion test results after 4 hours of salt spray: left photo: magazine boxes with stainless on the left and EN on the right; right photo: barrels with EN on top and stainless on bottom.
Specimens after 45 hours of salt spray: Figure 5 shows the test parts after 45 hours of salt spray, labeled as stainless steel or EN-plated. One-hundred percent red rust was observed on the stainless steel barrel, while there was no rust on the EN-plated barrel, except inside where there was no plating. There was extensive rust on the bolt head of the stainless steel receiver, while there was no rust noted on the EN-plated receiver. However, both receivers exhibited corrosion from the steel scope mount screws, which were not stainless, nor were they plated. Mechanically, the stainless steel action was beginning to show resistance. No corrosion was noted on the EN-plated receivers or bolts. Magazine box A showed no rust on the stainless steel finish and less than 1 percent rust on the EN-plated box. Box B showed no rust on the stainless steel finish and less than 2 percent rust on the EN-plated box. Box C exhibited no corrosion with either finish.


Figure 5: Corrosion test results after 45 hours of salt spray: in left photo EN is on top and stainless on bottom; in right photo stainless is left and EN right.
Specimens after 93 hours of salt spray (end of test): Figure 6 shows the test parts at the end of the test, after 93 hours of salt spray, labeled as stainless steel or EN-plated. As at 45 hours, 100 percent red rust was observed on the stainless steel barrel, while there was no rust on the EN-plated barrel, except inside, where there was no plating. The bolt head of the stainless steel receiver was 100 percent rusted, with extensive rust on the bolt handle. There was no rust noted on the either receiver except that from the steel scope mount screws. Mechanically, the stainless steel action was completely nonfunctional, while the EN finish action was still fully functional. No corrosion was noted on the EN-plated receivers or bolts. Magazine box A showed slight rusting on the stainless steel finish and about two percent rust on the EN-plated box. Box B showed slight rusting on the stainless steel finish and five–10 percent rust on the EN-plated box. Box C showed slight rusting on the stainless steel finish and less than one percent rust on the EN-plated box.


Figure 6: Corrosion test results after 93 hours of salt spray at the end of the test: on the left photo, barrels with EN on top and stainless on bottom; and in the right photo, magazine boxes with stainless on the left and EN on right.
The EN-plated 1155 steel gun significantly outperformed the 416R stainless steel gun in corrosion resistance as tested with neutral salt spray. The 416R stainless steel barrel began to show red rust within a few hours of being subjected to salt fog, where, at the end of the test at 93 hours, the EN-plated barrel exhibited no corrosion on the outside of the barrel where it was coated. On the inside of the barrels, where there was no EN coating, the barrels exhibited similar rusting conditions.
Oddly, the 416R stainless steel receiver performed as well as the EN-plated 1155 steel receiver. It was surmised that this was because the receiver was subjected to a different heat treat cycle than the barrel.
As far as the functionality of the firing mechanisms, the EN-plated gun continued to function, both in firing and safety, throughout the testing, whereas the stainless steel gun failed after approximately 70 hours of salt spray exposure.
The stainless steel magazine boxes, being made of the more corrosion resistant 304 stainless steel, outperformed the EN-plated magazine boxes, but not by the overwhelming margin that the EN-plated barrel outperformed the stainless steel barrel. The EN thickness on the magazine boxes was minimal, and it can be reasonably surmised that if they had been coated to a higher thickness, they would have performed more comparably to the 304 stainless boxes.
Cost savings were calculated at more than $17 per gun when using EN-plated steel instead of the 416R or 304 stainless steel. With nearly 11,000 stainless steel units produced each year, that adds up to $187,000 annually, with improved corrosion resistance and increased functionality.
Conclusions
While replacing stainless steel is not feasible in every instance, from these two case studies, it’s clear that EN-plated steel may be able to replace stainless steel in certain applications. It is the challenge of the industrious metal finisher or the visionary manufacturer to find new applications where this strategy might work and empirically determine its chances of success.
"The Electroless Deposition of Nickel-Phosphorus-Tungsten Alloys": This paper describes the successful electroless co-deposition of tungsten within a nickel-phosphorus matrix to yield several advantageous properties.
Originally published in the October 2015 issue.
RELATED CONTENT
-
Choosing and Troubleshooting Copper Electroplating Processes
Learn more on this inexpensive and highly efficient process.
-
Stripping of Plated Finishes
The processes, chemicals and equipment, plus control and troubleshooting.
-
Zinc Electroplating
Choosing the best process for your operation.


















