Novel Post Treatment of Nano-particle Reinforced Metallic Coatings
This paper discusses a novel post-treatment reinforcement process for both electroless and electrolytic nano-composite coatings which further improves the hardness of the coating (+40%), while also improving the ductility of the material. This technology is applicable to commercial baths for a large variety of electrolytic and electroless coatings. Interestingly, post-treated coatings retain performance at higher temperatures, suggesting improved oxide dispersion strengthening effects, potentially offering new coating options for high temperature applications.
#surfin
Editor’s Note: The following is a synopsis of a presentation given at NASF SUR/FIN 2019, in Rosemont, Illinois on June 3, 2019 in Session 7, Advances: Research. A pdf of this brief can be accessed and printed HERE; the complete Powerpoint presentation is available by clicking HERE. The SUR/FIN 2017 Powerpoint presentation referred to in the Abstract below (as a pdf) is available HERE.
ABSTRACT: Cirrus Dopant technology, previously discussed at SUR/FIN 2017, offers a means to deposit nanocomposite metal coatings without the implementation and process drawbacks of powder mixing. Recently, Cirrus developed a novel post-treatment process for both electroless and electrolytic nano-composite coatings reinforced with Cirrus Dopant which further improves the hardness of the coating (+40%), while surprisingly also improving the ductility of the material. This technology is applicable to commercial baths for a large variety of electrolytic and electroless coatings. Interestingly, post-treated coatings retain performance at higher temperatures, suggesting improved oxide dispersion strengthening effects, potentially offering new coating options for high temperature applications. This presentation discusses the process and latest results for nano-doping commercially relevant coating baths, including updated performance results for post-treatment of nano-composite coatings and oxide dispersion strengthening for electroless plating.
Featured Content
Introduction: Cirrus Materials Science Ltd., based in Auckland, New Zealand is an advanced coatings technology company. They develop expertise and apply novel, patented intellectual property to advanced, high performance coating challenges. One such technology is Cirrus Dopant, a means to commercially create high performance nano-composite coatings by introducing nanoparticle strengthening without the problems associated with nano-powders. Dopants are Cirrus’ foundation technology, which are now in their fourth product generation. The patented drop-in technology fits into many existing processes, and the latest dopants are customized for both compatibility with specific plating baths and to create specific coating properties. Dopants are available to codeposit zirconia, titania, alumina and ceria particles into a wide range of performance metal coatings. Typically, with these new dopants, less than 1% by volume of a dopant is required to develop enhanced coating properties.
Background: Mastip, a New Zealand company, is a global innovator in the plastic hot runner industry. Hot runners maintain the temperature of the molten polymer thereby improving molding efficiency. Material selection for the injection tips is critical to maintaining a flat thermal profile. Mastip offers several tip ranges, one using a high-cost tungsten carbide, and other lower-cost tips based on copper-beryllium. Copper beryllium tips are the most common, and the substrate must be protected by a thin wear-resistant protective coating, which maintains thermal performance.
However, tip erosion from emerging new injection-molded polymers (e.g., glass-filled polymers) lowers tip life and results in machine downtime. Currently 20 microns of a high phosphorus electroless nickel coating is standard and Mastip came to the University of Auckland and Cirrus looking to improve the tip life by about 40%. The University analyzed various non-dopant approaches and Cirrus evaluated dopant approaches to improve tip life. As the tips operate at a temperature between 200 and 350°C, one part of the investigation was to see how the temperature might affect hardness and erosion.
Results
Initial evaluations at the University demonstrated that, as expected, heat-treatment produced a harder but less wear-resistant coating using the high phosphorus electroless nickel bath. Simultaneously, Cirrus evaluated several doped coatings, the results of which are shown in Fig. 1, based on which, both the University and Cirrus proposed a change from high phosphorus to doped low-phosphorus coatings. Mastip accepted this finding and several demonstration tips were coated, reference tips were provided by their current supplier. Mastip independently performed a side-by-side comparison of the current and doped tips using their onsite injection molding machine and a highly aggressive glass reinforced polymer at an injection temperature of 240°C.
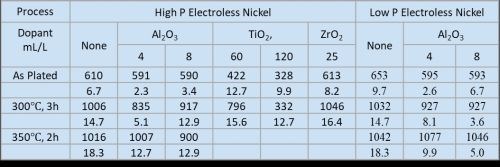
Figure 1 - Laboratory coating evaluation results.

Figure 2 - Comparison of wear between current ENiP and doped ENiP coatings.
The heat treated high phosphorus electroless nickel-coated tip developed accelerated wear between 400 and 500 injection cycles (Fig. 2(a,b)). However, the heat-treated, doped low-phosphorus coatings, increased wear-resistance. A doped low-phosphorus coating was more wear-resistant (Fig. 2 (c,d)). These real-world results were unexpected, with virtually zero erosion after 4000 cycles and suggested further research.
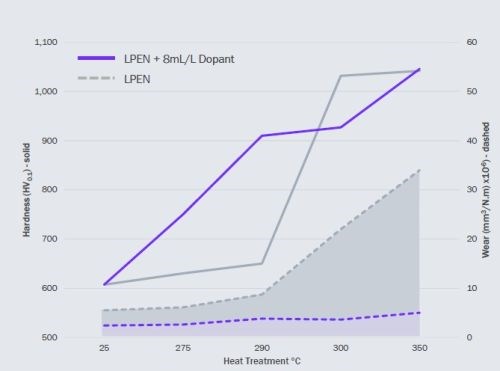
Figure 3 - Comparison of doped and undoped low-phosphorus ENi coatings heat-treated at various temperatures.
Figure 3 compares the hardness and wear resistance of doped and undoped low phosphorus electroless nickel coatings heat-treated at various temperatures. Here, the heat-treatment durations were 1 hr except for 350°C, where the samples were treated for 4 hr.
The main observations are that doped coatings, when heat-treated, become harder at lower temperatures than undoped coatings. The ultimate hardness, created by precipitation of nickel-phosphides, was about the same for doped and undoped coatings. Precipitation hardening occurs at all temperatures, but is accelerated above 300°C.
The low temperature hardening of doped coatings was of significant interest as the mechanism was unexplained at that time. However, of more interest to the Mastip application was the measured wear performance of the doped coatings. While the wear rate of an undoped low-phosphorus coating quadruples as precipitation hardening occurs, the wear rate on the doped coatings remains substantially unchanged.
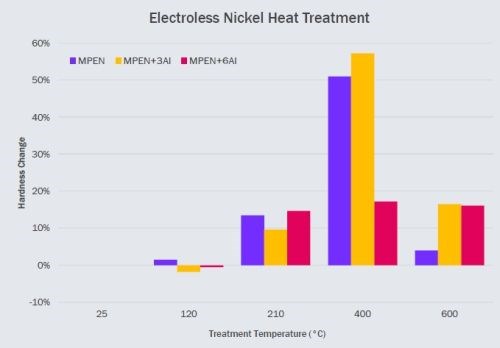
Figure 4 - Comparison of heat-treatment response versus doping levels.
We next examined the effect of doping level on the low temperature hardening of electroless nickel deposits. Here, a high concentration alumina dopant was used, thus the doping levels in (Fig. 4) cannot be directly compared with those from Fig. 3. The particular dopant used develops about 2.5 times the number of particles as a low concentration dopant. Consequently, a 3 mL/L in Fig 4 more or less equivalent to 8 mL/L from Fig. 3.
A medium phosphorous electroless nickel bath was used where the particle size developed by this high concentration dopant is also somewhat different. Hardness values, reported in Fig 4, are relative to as-plated hardness to highlight trends and eliminate variation between baths. We observed that a pure mid-phosphorus ENi coating hardens gradually with temperature, potentially due to strain hardening, until the temperature reaches between 300 and 400°C, where intermetallic precipitation significantly increases the hardness. At 600°C, the coating substantially loses its hardness improvement.
Coatings doped with low concentrations initially slightly soften, followed by an increase, where particle-modified strain hardening occurs. Note that the absolute hardness here is greater than that of an undoped coating. Intermetallic precipitation creates a significant jump in hardness. Finally, at 600°C, the intermetallic increase is lost but the residual strain hardness remains.
High dopant concentrations interfere with the intermetallic formation and there is only a slight increase in harness at 400℃. We have previously noted that particle generation and incorporation in electroless coatings is associated with the hypophosphate. Thus, the observed lack of precipitation hardening is expected. Once again, the hardness improvement is retained at temperatures above 600℃.
We next examined low temperature hardening of pure nickel electroplated coatings, see Fig 5. Here we plated 2.5 μm of nickel and doped nickel on mild steel substrates. We adopted a very low temperature heat treatment temperature of 120℃. Our previous results had demonstrated that a zirconia generating dopant produced excellent results with an electroplated nickel coating.
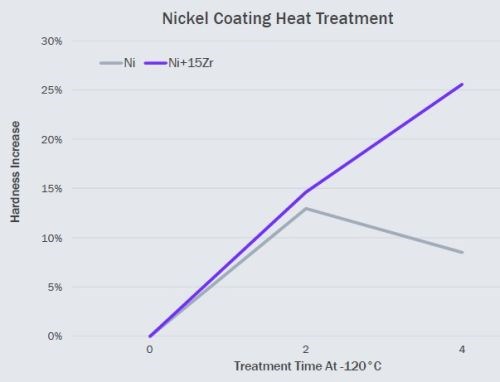
Figure 5 - Comparison of low-temperature heat-treatment hardness response of undoped and Zr-doped pure nickel electrodeposits.
In Fig. 5, once again hardness relative to the “as plated” hardness is reported to allow direct comparison of coatings with differing initial harnesses, here we see that initially, both doped and undoped coatings show strain hardening. Over time however, the undoped coatings lost their hardness improvements as grain growth relieves the strain. Doped coatings on the other hand, continued to harden.
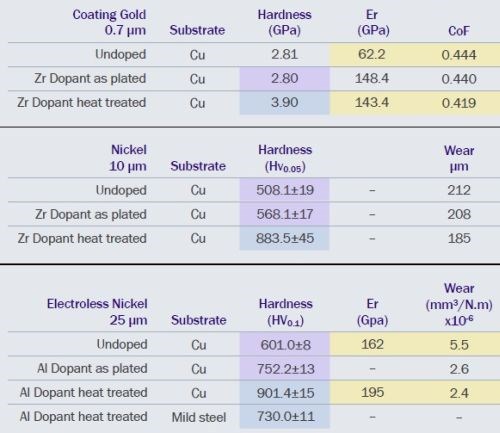
Figure 6 - Comparison of hardness, modulus and coefficient of friction for a range of coatings, doped and undoped.
In Fig. 6 we report results on a range of coatings deposited on two substrates to demonstrate low-temperature heat-treatment effects. Considering hardness, all doped coatings are initially harder than their undoped counterparts. Low temperature heat-treatment, at 200°C for 2 hours, further increases the hardness of doped coatings. Interestingly, the hardness values of low temperature heat-treated electroless nickel coatings on copper and mild steel substrates, are different. This supports a strain or work hardening strengthening mechanism; here differential thermal expansion between the substrate and the coating is the strain mechanism. Finally, although the coatings are harder, the reduced modulus, a measure of ductility, is higher an unexpected result. Further research is required. Nonetheless, the improved wear performance of the heat-treated coatings appears to be correlated with the improvement in reduced modulus.
General Observations
While the observations below are not all directly supported by the data presented above, we believe:
- Strain or work hardening created by differential thermal expansion between the costing and substrate is the strengthening mechanism, while not included in the results presented, we have observed hardening improvement when nickel on copper samples are heat treated at -165°C.
- Grain growth, even at low temperature, appears to soften undoped coatings despite strain hardening.
- At higher temperatures, both intermetallic formation and strain hardening have an impact, but highly doped coatings can reduce the rate of intermetallic formation.
- The retention of improved hardness above the expected re-melt temperatures has been observed and requires further investigation.
- Nanocomposite particle density may be one factor associated with suppression intermetallic particle formation, however we previously observed less compact coatings as the particle concentration increased.
- Nano composite particle size may be a factor, as we observe significant behavior differences with zirconia dopants (10 nm particles), and various alumina dopants (40-60 nm particles).
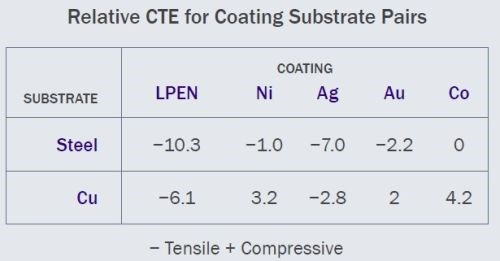
Figure 7 - Relative coefficient of thermal expansion for various coating/substrate combinations studied.
Hardening
As previously noted, hardening depends on both the substrate and coating. We postulate that the relative coefficient of thermal expansion (CTE) underlies the measurement improvements. In Fig. 7, we show the relative CTE for coating-substrate combinations which we have evaluated. Our observed results are closely correlated with the relative CTE with improvement greater with high relative CTE and less at lower. Doped silver is the exception here; heat treated coatings have softened. However, coating thickness may have impacted the outcome.
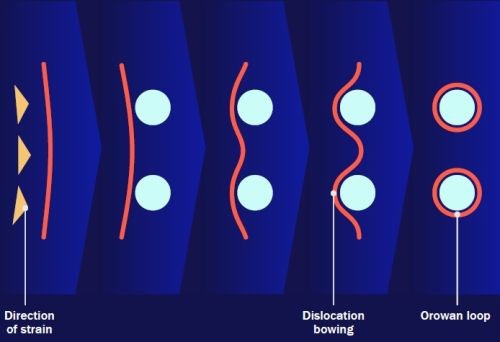
Figure 8 - Possible hardening mechanisms.
A suggested hardening mechanism for the doped deposits is shown schematically in Fig. 8. We have observed hardening operating at both low temperature heating and cooling, suggesting that matrix strain, created by differential thermal expansion of coatings versus substrate is a significant factor. Strain or work hardening is modified by particle presence as a dislocation wrap around the particles. We believe that these wrapped dislocations “artificially” shrink the interparticle distance and thus lock in the hardness improvement. We have also observed that copper substrates create a higher improvement than mild steel substrates, indicating that the relative expansion between the substrate and a thin coating contributes to improvements. The relative thinness of the coatings results in largely two dimensional rather that three dimensional induced strain Differences between the alumina and zirconia dopants may also demonstrate a particle dewatering influence, where particle shrinkage induces local strain.
We have observed some coating hardening effects at temperatures as low as 120°C. Thus, this process could well be applied to polymer substrates. The best results were seen for alloy coatings with excellent and vastly increased erosion resistance, which could provide important industrial benefits. The observed improvement in ductility of heat-treated doped coatings is important to wear performance. Finally, the improved high temperature performance of doped alloy costings offers benefits in certain applications.
Summary
A novel low temperature hardening that applies to doped coatings has been discussed. We have seen that the presence of dopant modifies the strain behavior of the coating, which improves the hardness. The hardening effect depends on the relative thermal expansion of the coating and substrate, which may be higher for thin coatings than thick coatings. The best results occur in electroless nickel and electroplated alloys.
About the authors

Dr. See Leng Tay is a Materials Engineer at Cirrus Materials Science Ltd. in Auckland, New Zealand. She completed her Ph.D. in 2016 at the University of Auckland, New Zealand under the “Bright Sparks” University of Malaya Overseas Scholarship Scheme. Her research areas include surface science and engineering, nanocomposite coatings, intermetallic compounds, lead-free solder, and she has published more than 30 journal articles and two patents in her field. See Leng Tay has won awards at numerous national and international exhibitions pertaining to her research work, including Gold medal in International and Technology Exhibition and Sparks Ideas Challenge 2015 (Commercial Ideas).

Chris Goode is a founder and Chief Technical Officer at Cirrus Materials Science Ltd., in Auckland, New Zealand. Chris holds an honors degree in engineering from the University of Western Australia. He holds over 30 patents and has extensive experience in multiple fields of engineering including telecommunications robotics and chemical engineering.
* Corresponding authors:
|
Chris Goode, CTO Cirrus Materials Science Limited C/- 203/16 Huron Street Takapuna Auckland, Auckland 0622 New Zealand Phone: +64 21 248 8853 Mobile: +64 21 248 8853 E-Mail: chris.goode@cirrusmaterials.com |
See Leng Tay, Senior Materials Engineer Cirrus Materials Science 5B Target Court Wairau Valley, 0627 New Zealand Phone: +64 22 095 2461 |
RELATED CONTENT
-
Smut and Desmutting
Question: I am new to this industry and have heard about smut and desmutting operations.
-
Copper Plating on Aluminum and Aluminum Alloys
How can I plate copper on aluminum?
-
Sizing Heating and Cooling Coils
Why is it important for you to know this?



















