Nanotechnology Offers Hard Chrome Alternative
Appears in Print as: 'Small Crystals, Big Benefits'
Nanotech cobalt alloy coatings offer hard chrome alternative
Aircraft landing gear, hydraulic actuators, gas turbine engines, helicopter dynamic components and propeller hubs all make use of hex chrome coatings. Recently, however, an electrodeposited nanocrystalline cobalt-phosphorus alloy, developed with funding from U.S. and Canadian defense partners, has come onto the scene. It has properties that are in many ways better than chrome, overcomes its environmental limitations and can offer improved performance and reduced life-cycle costs. Here’s the scoop on this new technology.
The Background
Electroplated engineering hard chromium (EHC) coatings 0.00025–0.010 inches thick are used extensively for imparting wear and erosion resistance to components in aerospace applications. Hard chrome deposits from hex chrome (Cr6+) baths are used in a variety of aircraft components, in both manufacturing and repair/overhaul operations.
Featured Content
In landing gear, for example, outer cylinder IDs are often chrome plated for wear and corrosion resistance. Internal chrome plating is most prevalent on landing gear components and hydraulic actuators. Electroplating lends itself to such applications, which would be difficult or impossible to coat using many of the line-of-sight (LOS) potential replacement processes developed so far.
Unfortunately, hex chrome’s toxicity has reduced its use significantly. OSHA, for example, recently reduced the permissible exposure limit for hex chrome and its compounds from 52–5 µg/m3 as an 8-hr time-weighted average. The rule also includes provisions for employee protection, such as preferred methods for controlling exposure, respiratory protection, protective work clothing and equipment, hygiene areas and practices, medical surveillance, hazard communication and record keeping.
In addition to the health risks associated with hex chrome, there are other process and performance drawbacks associated with use of EHC coatings. EHC plating processes generally have a relatively low electrolytic efficiency, resulting in low deposition rates compared to other plated metals and alloys. Moreover, the intrinsic brittleness of EHC deposits invariably leads to micro- or macro-cracked deposits. These cracks do not compromise wear and erosion resistance, but they are wholly unsuitable for applications where corrosion resistance is required. In these applications, an electrodeposited underlayer of a more ductile and corrosion-resistant material—usually nickel—must be applied.
As a result of these health and safety restrictions and process/performance drawbacks, there is tremendous pressure in the electroplating industry to find a more environmentally benign alternative to hard chrome. Technologies considered as alternatives include thermal spray, plasma vapor deposition, and other chrome-free materials applied by electrolytic or electroless plating techniques.
Over the last 10 years, tungsten carbide-cobalt (WC-Co) and similar materials applied using high-velocity oxygen-fuel (HVOF) thermal spray have undergone extensive demonstration/validation testing as part of the U.S. Department of Defense Hard Chrome Alternatives Team (HCAT) program. These materials have generally been accepted as suitable alternatives for hard chrome within the North American aerospace industry and for other low-volume, high-added-value LOS coating applications. For coating applications requiring non-line-of-sight deposition (NLOS) and/or high-volume, low-value-added production, however, it’s generally believed that only electroplating technologies will be suitable and/or cost-effective.
Most of the electroplated coating alternatives investigated so far have been based on nickel alloys, including both electroless and electrolytic materials. Because nickel is listed by the EPA as a priority pollutant and is considered to be one of the 14 most toxic heavy metals, coatings containing nickel represent a short-term solution at best. Therefore, a non-nickel-based electroplating technology would be a practical, environmentally acceptable alternative for NLOS coating applications.
Enter Nanotech
Nanotechnology is a relatively new field that deals with the design of extremely small structures having critical length dimensions on the order of a few nanometers. Nanostructured materials—materials with an ultra-fine average grain size usually less than 100 nm—were initially introduced as interfacial materials about 20 years ago. The main characteristic of these materials is an enhanced volume fraction of the interface component (the volume fraction of atoms associated with grain boundaries and triple junctions). This becomes significant when average grain size decreases below 100 nm. Having such a large fraction of atoms located at the interfacial defect structure causes changes in many mechanical, physical and chemical properties of nanocrystalline materials.
The first systematic studies on the synthesis of electrodeposited nanocrystalline materials attempted to optimize certain properties by deliberately controlling the volume fractions of grain boundaries and triple junctions in the materials. Since then, many nanocrystalline metals and alloys have been produced by electrodeposition, including pure nickel, cobalt, palladium and copper; binary alloys such as nickel-iron, nickel-phosphorus, zinc-nickel, palladium-iron and cobalt-tungsten; and ternary alloys such as a nickel-iron-chromium material.
Another such material is Nanovate CR, an electrodeposited nanocrystalline cobalt-phosphorus alloy developed and demonstrated by our company with funding from U.S. and Canadian defense partners. The electrodeposition process can be used in both LOS and NLOS applications, and the material can be viewed as part of an overall strategy to replace currently used EHC processes while significantly improving performance and reducing life-cycle costs.
As shown in Table 1 (below), the Nanovate CR process offers significant improvements over EHC. Like EHC, the material is produced by electrodeposition. It therefore represents a drop-in alternative technology that is fully compatible with the current hard chrome electroplating infrastructure and is well-suited for application to both LOS and NLOS surfaces. Unlike EHC, the process uses no constituents on U.S. EPA or other lists of hazardous materials, nor does it generate hazardous emissions or by-products.
|
Table 1: Comparison of Nanovate CR and EHC Processes
|
||
|
|
Nanovate CR
|
EHC
|
| Deposition Method |
Electrodeposition
|
Electrodeposition
|
| Applicable Part Geometries |
LOS and NLOS
|
LOS and NLOS
|
| Efficiency, % |
85–95%
|
15–35
|
| Deposition Rate, iph |
0.002–0.008
|
0.0005–0.001
|
| Appearance |
Free of pits, pores or cracks
|
Microcracked
|
| Microstructure |
Nanocrystalline (avg. grain size = 5–15nm)
|
-
|
| Emission Analysis |
Below OSHA limits
|
Cr6+
|
Use of the nanotechnology process also results in significant reductions in energy consumption and increases in throughput. Overall plating efficiency is approximately 90%, compared to less than 35% for EHC. Further, Nanovate CR has a deposition rate ranging from 0.002–0.008 iph, depending on current density, versus the 0.0005–0.001 iph deposition rate typically seen with ENC processes.
Properties
Visually, nanocrystalline cobalt-phosphorus coatings are uniformly smooth and shiny, similar to EHC. Microscopically, deposits are fully dense structures free from pits, pores and microcracks.
Metallurgically, the material exhibits a hexagonal close-packed (HCP) crystal structure, the equilibrium structure typically found in conventional cobalt at room temperature. Unlike conventional cobalt, however, the material has an average grain size in the range of 5–15 nm. Testing shows that an average grain size in this range results in an optimal combination of strength and ductility. Table 2 (below) compares the properties of nanocrystalline cobalt alloy and EHC.
|
Table 2: Comparison of Nanovate CR and EHC Properties
|
||
| Nanovate CR | EHC | |
| Hardness, HVN | 530–600 (as deposited) | Min 600 |
| 600–680 (heat treated) | – | |
| Wear Volume Loss, mm3/Nm | 6–7 × 10-6 | 9–11 × 10-6 |
| Coefficient of Friction | 0.4–0.5 | 0.7 |
| Pin Wear | Mild | Severe |
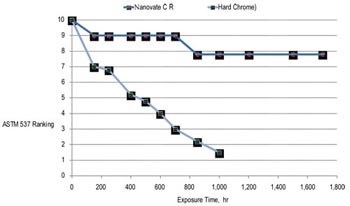
| Corrosion Resistance* | 8 | 2 |
| Hydrogen Embrittlement | Pass with bake | Pass with bake |
| *ASTM B 537 protection rating after 1,000 hr salt-spray exposure per ASTM B 117 | ||
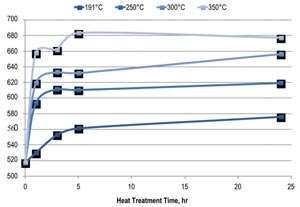
Nanocrystalline alloys such as Nanovate CR display significant increases in hardness and strength relative to their coarser-grained, conventional counterparts. Through a solid solution hardening mechanism, microhardness values typically range from 530–600 VHN.
A further increase in hardness can be obtained by annealing the as-deposited material. A short heat treatment process results in microhardness increases of more than 150 VHN.
Nanovate CR also has improved wear and lubricity relative to EHC. The material exhibited less wear loss than EHC in pin-on-disk sliding wear testing. Wear loss of the mating material—in this case, an alumina ball—was also less severe, indicating that nanocrystalline cobalt-phosphorus has a lower coefficient of friction than EHC.
Corrosion resistance in salt-spray testing is also improved. In a comparison of Nanovate CR and other hardfacing materials after 1,000 hr of exposure in a salt-spray environment per ASTM B 117, the material’s ASTM B 537 protection rating decreased to only 8, compared with a rating of less 2 for EHC. Further, the nanocrystalline deposit was 50% thinner than the EHC and HVOF coatings used in the test.
Another important consideration in aerospace plating is the potential for hydrogen embrittlement in high-strength steel components. The high plating efficiency of the Nanovate CR process leads to significantly less hydrogen generation at the cathode compared with EHC processes, thus minimizing likelihood of hydrogen uptake and subsequent embrittlement of susceptible materials. Hydrogen embrittlement tests conducted in accordance with ASTM F 519 indicate that standard hydrogen embrittlement relief baking procedures for EHC can be applied to nanocrystalline deposits to fully eliminate any risk of embrittlement.
Adhesion of nanocrystalline cobalt-phosphorus deposits has been evaluated on a number of aerospace substrate materials. In bend tests conducted in accordance with ASTM B 571, deposits showed no signs of peeling or delamination at low (10×) magnification. In testing in accordance with ASTM B 553, samples coated with Nanovate CR were exposed to thermal cycling involving submerging the samples into liquids nitrogen for one minute followed by submersion in hot (90 ºC) water for one minute. After 30 thermal cycles no delamination occurred and the displacement of the coating relative to the underlying substrate was substantially zero.
Acknowledgements
The authors gratefully acknowledge support from the U.S. DoD Strategic Environmental Research and Development Program (SERDP) and Environmental Security Technology Certification Program (ESTCP), and Technology Partnerships Canada.
RELATED CONTENT
-
Blackening of Ferrous Metals
The reasons for installing an in-house cold blackening system are many and varied.
-
Sizing Heating and Cooling Coils
Why is it important for you to know this?
-
Stripping of Plated Finishes
The processes, chemicals and equipment, plus control and troubleshooting.