Choosing the Best Electrolytic Nickel Strike
Now that you have decided that an electrolytic nickel strike would benefit your process, how do you choose the best one for your application? Coventya’s Brad Durkin explains.
In Part 1 of this article in the November issue, we provided a summary of the reasons for utilizing strike chemistry on the plating line. Now that you have decided that an electrolytic nickel strike would benefit your process, how do you choose the best one for your application? There can be some confusion on the choices available, but now we will focus on the differences between the various nickel strike types to make the selection process easier. The types of nickel strikes and the appropriate use include:
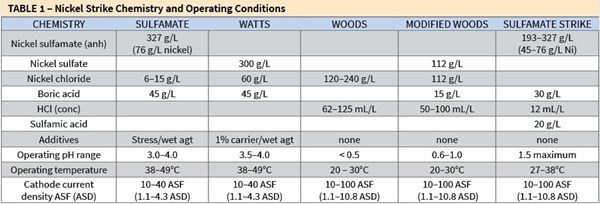
- Watts for catalyzing non-catalytic substrates and for corrosion-barrier underlayer; pH 3.5–4.0
- Sulfamate for catalyzing non-catalytic substrates and for corrosion-barrier underlayer; pH 3.0–4.0
- Woods for activating high chromium and nickel alloy types; pH <0.5
- Modified Woods for activating and catalyzing; pH >1
- Sulfamate strike for activating and catalyzing; pH 1.5
Tables 1 and 2 provide an overview of the chemistry and operating conditions, and a selection guide as to which type might provide the best performance for given applications. Because most electrolytic strikes are applied for the activation of difficult substrates, comparing and contrasting the Woods, modified Woods and sulfamate strikes merit some additional discussion and perspective. The following discussion supplements the information contained in Table 2.

Sulfamate nickel activating strike. This is the least commonly used today, but it offers some viable advantages over the other two formulations. First, nickel sulfamate is a more expensive source of nickel and is not readily available compared to nickel sulfate. However, sulfamate nickel activating strikes offer higher purity deposits and lower stress that can be ideal for some critical applications, including those that require bending and ductility. A strike formulation of this type allows improved and successful activation in barrel plating applications, which is difficult for other nickel strikes to accomplish.
This chemistry is better suited for multi-metal parts and components; there is less potential for the electrolyte to attack the many substrate types. Since the formulation’s operating pH is higher, this chemistry is less corrosive overall and easy to purify with carbon if required, and the solution can be dummy-plated to remove impurities, unlike with other activating nickel strikes. It can utilize traditional SD nickel rounds in titanium baskets, which is more common for electrolytic nickel plating.
Because of the higher cathode efficiency and lower acidity compared with the other two activating strikes, the sulfamate chemistry does not tend to increase the nickel metal concentration that is detrimental and decreases the activation potential of those lower-pH formulation types. Additionally, where poor rinsing exists on process lines, dragging this chemistry into an electroless nickel bath does not cause serious implications.
On the downside, despite the strike’s long life, its make-up cost is high, and it is not as consistent an activator on many nickel-bearing stainless steel or alloys with nickel, so justification to install this type of operation can be difficult. Additionally, sulfamate anion degrades over time into ammonium species that can trigger some waste-treatment concerns for some applicator facilities with mixed rinse streams.
Woods nickel activating strike. This chemistry has been around for numerous decades, and many variations have evolved from the original, but it was designed to be an activator/strike for providing adhesion on difficult ferrous-based substrates. There can be much debate on the formulation, as shown in some sources that are not all inclusive of variations:
AMS Metals Handbook:
• 240 g/L NiCl2 6H2O + 250 mL/L HCL (37 percent)
ASTM B 656:
• 240 g/L NiCl2 6H2O + 320 mL/L HCL (37 percent)
• Anodic 30-60 seconds, then cathodic 2-6 minutes
Other source variations/publications:
• 240 g/L NiCl2 6H2O + 125 mL/L HCL (37 percent)
• 180 g/L NiCl2 6H2O + 160 mL/L HCL (37 percent)
• 120 g/L NiCl2 6H2O + 80–00 mL/L HCL (37 percent)
Regardless of variations, there are some real benefits of incorporating this type chemistry into a process line in which there might not be enough room for both an acid activation tank and electrolytic strike. It works well. Early versions of this strike were operated as an anodic activation (reverse-etch) followed by a cathodic current to strike and provide a very thin layer of nickel to displace surface oxides.
As a result, and despite the contamination from reverse-etching, this chemistry is excellent for activation on nickel-bearing stainless alloys, which include a wide range of substrate types. The uniqueness for use as a reverse-etch on some alloy types offers advantages. More importantly, it is very inexpensive to make up, so many applicators think of it as a throw-away type of activation chemistry.
In contrast to sulfamate, this chemistry is highly acidic and corrosive, and a result of its use is prone to metallic contamination. It is common to experience immersion copper deposits that cause poor adhesion on some substrate types depending on operating conditions. Overall, metallic contamination cannot be dummy-plated from this high acidic chemistry, thus causing a short life while the solution must be more frequently replaced and waste-treated. Also, because of the very high acidity, electrolytic nickel anodes are utilized, which are offered in many different grades and impurity levels. Solution nickel metal increase is common, which reduces the ability of the strike to activate some types of substrates. Unless anodes are removed at the end of each production shift, the constant removal of some solution is required to maintain the inefficiency of the chemistry.
This chemistry is not good for activating barrel-plated parts due to the low current densities required where the solution tends to attack some substrate types instead of activating them. These formulations are not designed to provide any corrosion-barrier layers or to encapsulate porosity, or should not be dragged into electroless nickel chemistry without rinsing.
Hybrid modified Woods nickel activating strike. This chemistry evolved, in a sense, as a son to father Woods strike. With a need to gain good activation on nickel-bearing alloys, while still taking a more traditional electrolytic strike approach, this type fills a gap. Realizing some of the negatives experienced with the traditional woods formulation and wanting also to achieve some performance benefits offered from the sulfamate nickel strike but at a lower cost, the modified Woods hybrid formulation was born. Except for the very high nickel-bearing stainless steel alloys (>10 percent nickel), this strike formulation has a good activating potential for a wide range of difficult alloys. Additionally, this is not as acidic, reducing the potential attack of iron-chromium alloys. This chemistry results in some ideal applications of activation in barrel plating processes. Additionally it can be purified by dummy treatment for a longer tank life that fills a niche between that offered by the Woods and sulfamate strike formulations. The downsides include the fact that there are more components to analyze and maintain; the analysis for nickel coming from nickel chloride, nickel sulfate and overall acidity coming from chloride and boric acid concentrations can be a little more involved but straightforward. Because of the high chloride in the formulation, this does not offer good under-layer barrier deposits compared to the sulfamate but can work well for catalyzing some substrate types.
When it comes to catalyzing applications or providing good under-layer barrier deposits, either the Watts formulation or the sulfamate formulations work well. However, where improved higher corrosion-resistant deposits are required, sulfamate deposits offer higher purity, and their chemistries are not based on high concentrations of chloride or sulfur-bearing grain-refining additives like Watts. Of course, the Watts approach will provide less-costly chemistry formulation options, but when applications require any element of low internal stress (without additives) or high ductility (bending), the sulfamate nickel approach is going to be the winner in most cases.
Often, applicators will add bright nickel additives to these systems that work well or some that might not designed to be used in these chemistries, causing failure. For simple, high-build (thickness) applications, either type of deposit will meet the demand, and both formulations operate in similar fashion from an equipment perspective.
Brad Durkin is the director of international product management at Coventya. Visit coventya.com.
Related Content
Conductivity and Plating
Eric Xie of PAVCO Inc. discusses conductivity when considering the combination of plating metal deposits, passivates and topcoats/sealers.
Read MoreProducts Finishing Reveals 2024 Qualifying Top Shops
PF reveals the qualifying shops in its annual Top Shops Benchmarking Survey — a program designed to offer shops insights into their overall performance in the industry.
Read MoreThree Tips for Optimizing Surface Prep for Plating
By identifying oil slicks, performing in-line tests and observing tank appearance, many problems are solved regarding surface preparation in the plating process.
Read MoreFinisher’s ‘Top Shop’ Status Attracts Business
This competitive California finisher made it a goal to become a PF Top Shop. After earning the recognition, the company experienced an immediate increase in business and a challenge to obtain certifications.
Read MoreRead Next
Selecting a Spray Applicator for Liquid Coatings
Struggling to pick the perfect spray gun for your finishing ops? Explore spray applicator types, advantages and applications for optimal transfer efficiency and finish quality.
Read MoreEmbracing the Suck: Do You and Your Team Have What it Takes to Suffer?
The difference between athletics and business may not be not as vast as it seems — after all, both carry out lessons both important and necessary to building grit, enduring crises and, if you’re lucky, coming out on top.
Read MoreDoing More with Less: Why Efficiency and Durability Are Redefining Sustainability in Surface Finishing
The conversation around sustainability is evolving — and in surface finishing, how efficiently a coating is applied and how long it lasts matter more than ever.
Read More
























