Nanotech Silver Stops Microbes
Old, new technologies combine to reduce hospital infections
Hospital-related infections are a serious problem. The U.S. Centers for Disease Control (CDC) estimates that 2 million hospital patients a year acquire such infections. Approximately 100,000 of those afflicted die, making hospital-related infections the fourth-leading cause of death in the United States.
In addition to the personal toll there’s an economic price to be paid as well. Hospital-related infections cost an average of $57,000 per patient to treat. The added cost to hospitals, patients and insurance carriers for extended care and treatment is $28–30 billion annually.
Featured Content
About half of hospital-related infections are associated with catheters and other medical devices that provide a support surface for organisms to move into deeper tissue. Medical science has discovered that such infections are caused by formation of biofilms on the device surfaces. Biofilms are formed when rapidly mutating versions of free-floating bacteria attach to a surface. They are very difficult to get rid of and can result in serious blood infections, organ failure and death. The key to preventing biofilms is to stop bacteria from colonizing on the surface of medical devices.
Enter a new type of surface treatment that combines a very old anti-microbial material—silver—with the latest in nanotechnology can combat this infection mechanism and minimize the chance of infection in some types of surgeries. Developed by AcryMed Inc. (Portland, OR), SilvaGard silver nanotechnology can render existing medical devices impervious to infection-causing bacteria without dimensionally changing the device or chemically altering the treated surfaces.
Silver History
According to AcryMed, the anti-microbial properties of ionic silver have been known for centuries. In ancient Egypt and Greece, silver coins were dropped in fountains and silver goblets were used for drinking water to keep it potable.
Silver was also a popular antimicrobial well into the 20th century. One of its most widespread uses was silver nitrate drops placed in the eyes of newborns to prevent infections that could cause blindness. With the discovery of antibiotics, however, silver was no longer used extensively as a remedy for infection.
But unlike antibiotics, which are designed to fight specific microorganisms, ionic silver simultaneously attacks multiple sites in a cell to prevent it from reproducing or kill it. Pathogenic cells cannot mutate fast enough to avoid the attack, and as a result there are no known silver-resistant bacteria among medically relevant strains. It is this property that has caused doctors to consider use of silver technology for medical devices.
AcryMed says traditional silver antimicrobial treatments for medical devices fall into two broad categories: direct
incorporation and coating. Direct incorporation adds small particles of silver or silver salts directly into the device as it is being formed. It’s limited to non-metallic devices—in particular, plastic materials that are thermoformed or polymerized into the final device. Examples include wound dressings, catheters, tubes and trays.
Silver coatings, of course, can be produced by electroplating, by plasma- or sputter-coating, or by incorporating silver particles or salts into a coating which is ionically coupled or “painted” onto the outside of the device. All these methods change the surface properties and dimensions of the device.
Nanotech Silver
According to AcryMed, the SilvaGard process involves neither direct incorporation nor surface coating, and can be used to treat virtually any medical device. The process deposits discrete silver nanoparticles uniformly but discontinuously (on a submicroscopic, nearly molecular level) onto and into device surfaces.
Company president Jack McMaken says the process really is different than existing plating or organic coating processes. “It’s hard to put this into the same category as any conventional coating process,” he says. “It’s not really a coating at all—it’s really more of a surface functionalization.”
Despite this, operation of the process as explained by McMaken does not sound radically different from, for example, a conventional electroless plating process.
“We develop a chemistry that includes a source of silver ions—typically in the form of silver nitrate,” he says. “We also add a source of anions. We’ve used several types, including saccharinate and acetate anions. The formula also includes surfactants and other additives.
“All of the constituents are dissolved, and the solution is brought up to temperature because chemical processes generally go faster when they’re hotter. The I-Flow catheters (see below), for example, are processed at about 45°C.”
Once the bath is ready, items to be treated are immersed and a chemical initiator is added to start the reaction. “The initiator is like a catalyst except it’s consumed,” McMaken says. “It begins to take the silver ions out of solution and reduce them to metallic silver.
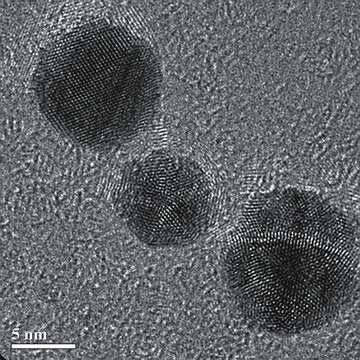
“This happens pretty much one atom at a time, and these single atoms try to attach to any surface in contact with the solution,” he explains. “As one atom attaches, it draws other atoms close to itself. The result is an agglomeration of atoms up to about 25,000 atoms, which is a nanoparticle. Nanoparticles are about 10 nm in diameter, and they’re firmly adhered to the surface of the part being treated.”
McMaken says size of the nanoparticles is self limiting. “For some reason we don’t quite understand, they really don’t grow much past that size,” he says. “They stop growing and instead go on to form another nanoparticle.”
Nanoparticle growth takes place over a period of minutes or hours, depending on the loading of nanoparticles desired on the surface being treated. “There can be a few nanoparticles, or there can be a lot depending on the antimicrobial efficacy we are looking for,” he explains.
“For things like the catheters, which might be in the body for several days, we look for a coating weight of about 8 μg/cm2. For permanent implants, we may put up to 100 μg/cm2 on there.”
The process can be aqueous or solvent-based depending on end use and substrate material being treated. The amount of nanoparticle silver actually deposited is controlled by adjusting the silver concentration and temperature of the solution as well as the dwell time in the solution. Treated devices are removed from the solution, rinsed thoroughly and dried, McMaken says.
In the body or on exposure to air, the outer layer of silver in each nanoparticle oxidizes to form a monolayer of silver oxide on the outside of the nanoparticle. The silver oxide slowly dissolves after the device is applied, forming ionic silver which is then available to attack and kill microbes.
Effective Treatment
The treatment has been used effectively on a variety of materials commonly used for medical devices. “We’ve done this with several dozen materials, and we haven’t found one yet that doesn’t work well,” McMaken says. “We have a couple of contracts on metal substrates for orthopedic and sensor applications. Metal substrates we’ve treated include stainless, titanium, chromium-aluminum alloys and copper. Getting metal nanoparticles to stick to metal is no problem. The harder thing is to get it to stick to a non-polar material such as polypropylene.”
Other materials treated thus far include glass, ceramics, silicon, and an array of fibers and polymers including cotton, rayon, polyester, polyethylene, polyurethane, polysulfone, nylon, silicone, polyimide, PEEK, PTFE and polycarbonate. Treatment leaves a light amber coloration visible on some materials.
McMaken believes the process would not be a huge leap for a finisher used to working in the medical market or providing services in other industries with rigorous process control and product quality requirements. “Once you have the formula, it’s simply a matter of watching your Ps and Qs—getting the right chemicals in there in the right order, and keeping tabs on the process,” he says.
“It requires fluid handling on the front end and the back end, temperature control, and some pretty rigorous quality control on the chemistry,” he continues.
“What we primarily assay for is silver loading, and we do that on the basis of micrograms per square centimeter, so you need something like an inductively coupled plasma or flame atomic absorption spectrophotometer to get down to those levels.
Another aspect of the process that will be familiar to finishers is waste treatment. “In the reaction we consume only a very small portion of the silver in solution,” McMaken says. “So you wind up with spent solution with some high percentage of what you initially put in there still in it. We’re currently working with a precipitation process to reclaim metal from spent solutions.”
McMaken says the process is flexible enough to be tailored to the demands of a wide variety of part sizes and production requirements. “We have potential customers, for example, with parts that might be 3 ft long and weigh 10 lb, and they only want to treat part of them. So there’s masking involved, and there’s fixturing, and larger tank sizes and fluid volumes,” he says.
“At the other end of the spectrum, we have a customer with a product consisting of three small pieces. The product sells for about $1.25, but they sell tens of millions of them. So they want a totally automated treatment process for all three parts, and they’ll probably treat maybe 250,000 pieces at a time.”
First Use
Initial U.S. Food and Drug Administration clearance for a device using the silver nanotechnology was given to I-Flow Corp. (Lake Forest, CA) for its On-Q SilverSoaker regional anesthesia delivery catheters. The devices are used by surgeons immediately after surgery to treat pain. They are inserted into the incision or somewhere near it before the surgeon closes the incision site, then hooked up to a system that delivers a local anesthetic. They can be left in the body up to five days after surgery.
According to I-Flow VP Orlando Rodrigues, the product is analogous to a soaker hose, which uses many small holes to provide watering to a specific area of a lawn. “On Q catheters consist of a filament inserted into the center of the hollow catheter,” he says. “The filament’s multiple holes allow us to get equal fluid flow along the length of the device without a lot of pressure. Catheters come in sizes from 2.5 to 10 inches long.”
Rodrigues says about 20 million U.S. surgeries each year could benefit from use of the technology. About 95% of those patients are still receiving narcotics for pain relief, and there’s been a learning curve to get surgeons to accept the catheter as an alternative for alleviating post-surgery pain.
“Early on, we found out surgeons were worried about infection—they’re leaving a foreign body in the surgical site, and they thought it increased the risk of infection,” he says. “So we went out and found the silver technology, which allows us to allay those concerns.”
The catheter portion of the device is a medical-grade nylon tube. It receives no special finish or surface treatment before the SilvaGard treatment, which is performed by AcryMed.
Rodrigues says his company sold about $70 million worth of the On Q catheters last year, and he sees a big potential up-side as more surgeons become aware of the product and its potential benefits. “In the U.S. alone, there’s probably about a $3 billion annual market,” he says.
RELATED CONTENT
-
Copper Plating on Aluminum and Aluminum Alloys
How can I plate copper on aluminum?
-
Cleaning, Pretreatment to Meet Medical Specs ISO 13485 or FDA 21 CFR820
Maximilian Kessler from SurTec explains new practices for industrial parts cleaning, metal pretreatment and decorative electroplating in the medical device industry.
-
Gold and Silver Plating Basics
An overview of precious metal electroplating processes.