Toxic Heavy Metal Free Stabilized Electroless Nickel Processes
This paper will introduce an advanced alternative to Pb and Cd-free electroless nickel processes, which is completely free of toxic heavy metal (THM) stabilizers. The process and deposit characteristics will be discussed and compared to the traditionally Pb stabilized electroless nickel.
#research #pollution control #masking
Abstract: The ELV/RoHS/WEEE directives have imposed restrictions on certain hazardous substances used in manufactured goods, which can pose harm to consumers and potentially cause a pollution risk to their environment after end-of-life. From the substances mentioned in these directives lead (Pb) and cadmium (Cd) which are used in electroless nickel fall under restriction. This paper will introduce an advanced alternative to Pb and Cd-free electroless nickel processes, which is completely free of toxic heavy metal (THM) stabilizers. The process and deposit characteristics will be discussed and compared to the traditionally Pb stabilized electroless nickel.
Featured Content
1. Introduction
Electroless nickel plating is an important and well established process, which is used in a wide spectrum of applications e.g. the oil and gas, automotive, electronics, white goods, aerospace, construction and textile industries.
The electroless nickel deposit provides an outstanding uniform coating thickness, which is able to improve the functionality of the plated parts by providing wear and corrosion resistance, excellent hardness and also abrasion resistance.
Traditionally, Pb was the commonly used stabilizer in electroless plating baths as a most effective catalytic poison for electroless nickel deposition. The addition of the stabilizer in the ppm range to the electroless nickel plating solution prevents spontaneous decomposition of the metastable plating bath. Cd has been traditionally used as an additive in the medium phosphorus electroless nickel baths to enhance the brightness and promote leveling.
ELV, RoHS and WEEE directives restrict the content of Pb to <0.1% and of Cd to <0.01% by weight in the coating. In order to avoid the necessity of tight analytic control of plated deposits, efforts have been undertaken to eliminate the use of Pb and Cd from electroless nickel plating completely.
In this context, Pb and Cd-free electroless nickel processes were developed and introduced. Nevertheless, the state-of-the-art electroless nickel plating processes still contain other toxic heavy metals as a stabilizer system. Such metals are not restricted by legislations today, but might be so in the future as there is a general demand for minimizing environmental impact of industrial processes.
An advanced generation of electroless nickel processes, which is free of Pb, Cd and toxic heavy metal stabilizers was introduced recently to the market. A process designed for the deposition of high phosphorous electroless nickel coatings (> 10 % phosphorus incorporation) was introduced in 2012 and a process generating medium phosphorous electroless nickel coatings (6 to 8 % phosphorus incorporation) was introduced in 2013. The challenge for this new technology is to offer a sustainable answer to the market with equivalent or better deposit and process properties compared to the traditionally stabilized electroless nickel processes.
2. Experimental
The THM stabilizer free electroless nickel processes have similar bath operating conditions as traditionally Pb stabilized electroless nickel processes. All plating tests were made according to internal technical data sheet operating conditions (1, 2).
The old generation electroless nickel processes using Pb and Cd often contain strong complexing agents like EDTA, in order to prevent the precipitation of nickel phosphite. Addition of EDTA results in very difficult waste water treatment. The new generation THM stabilizer free electroless nickel processes have been developed without use of EDTA.
The stress of the deposit was tested according to an internal standard operating procedure by using stress strips (3).
The nitric acid test of the deposit was performed according to an internal standard operating procedure (4).
3. Results and Discussion
3.1 Solution life and deposition rate
One of the important features of autocatalytic nickel solutions is the solution life, which is the time the solution can be used to produce deposits within acceptable quality requirements. The solution life is usually measured by the “metal turnover” (MTO). One metal turnover is achieved when the quantity of nickel contained in a new bath is converted into NiP deposit. An electroless nickel process operating at 6 g/l nickel has achieved one MTO when 6 g/l nickel has been deposited. High phosphorus electroless nickel processes suffer especially from very limited solution life. Production experience has shown that it is possible to extend the bath life from approx. 5 MTO to 7 MTO with the new generation high phosphorus electroless nickel process. Furthermore, the deposition rate of THM stabilizer free high phosphorus electroless nickel is higher compared to Pb stabilized high phosphorus electroless nickel processes (Fig. 1). The higher deposition rate as well as the longer operating life of the new generation electroless nickel process has major economic advantages for the applicator. The plating time is reduced, the make-up cost is reduced and the process bath is discarded less frequently which decreases the cost for waste treatment.
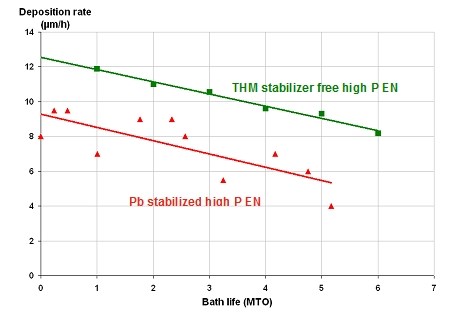
Figure 1. Deposition rate vs. bath life for THM stabilizer free high phosphorus electroless nickel and Pb stabilized high phosphorus electroless nickel.
3.2 Stress
A very important property of the electroless nickel deposit is the internal stress, which increases with bath age. For high phosphorus electroless nickel plating processes the deposit starts with compressive stress and becomes tensile over the bath life. This property affects the adhesion of the finished layer especially at high deposit thickness and the high internal stress can lead to formation of pitting (5). When tensile stress is observed the solution is typically discarded. Pb and Cd-containing high P processes turn from compressive to tensile stress after 4-5 MTOs, the new THM stabilizer free electroless nickel generation stays compressive for greater than 6 MTOs (Fig. 2).
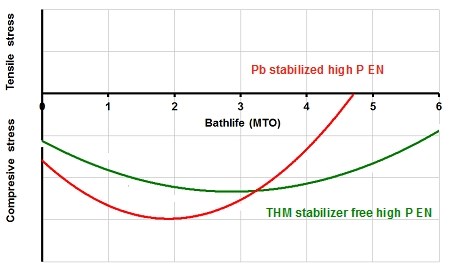
Figure 2. Internal stress vs. bathlife for THM stabilizer free high phosphorus electroless nickel and Pb stabilized high phosphorus electroless nickel.
The traditionally stabilized medium phosphorus electroless nickel deposit exhibits tensile stress. THM stabilizer free medium phosphorus electroless nickel provides close to neutral stress over the complete bath life (Fig. 3).
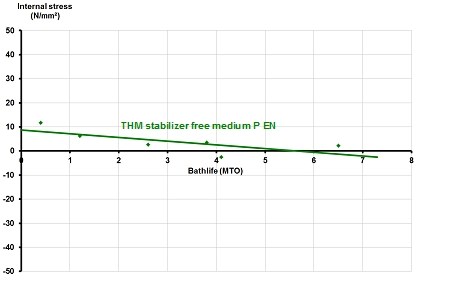
Figure 3. Internal stress vs. bathlife for THM stabilizer free medium phosphorus electroless nickel.
3.3 Nitric acid test
A nitric acid immersion test is commonly used in the industry as a fast, qualitative test to determine if the electroless nickel deposit provides medium or high phosphorus incorporation. An electroless nickel deposit with medium phosphorus content does not pass the 30 s dwell time in 65% nitric acid without discoloration. Traditionally stabilized as well as THM stabilizer free high phosphorus nickel deposits pass the nitric acid test. The results of investigations indicate that the new generation of electroless nickel processes shows no discoloration at an average of 3 minutes (Fig. 4).
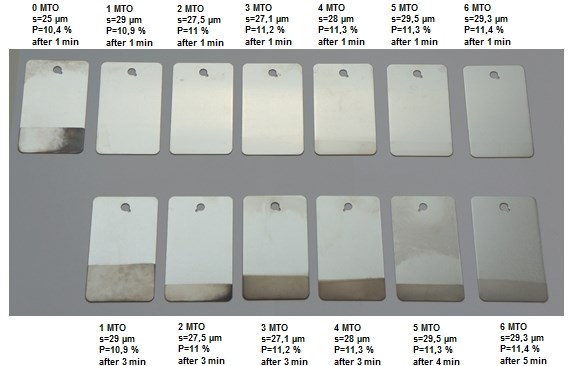
Figure 4. Results of nitric acid test for THM stabilizer free high phosphorus electroless nickel over the complete bath life.
3.4 Corrosion resistance
High phosphorus electroless nickel is used as a barrier coating against corrosive environment. Heavy metal stabilisers, like Pb can be a reason for poor corrosion of electroless nickel coatings, especially when the bath loading during production was too small or agitation was not appropriate. Lead is co-deposited in the electroless nickel deposit, which can result in pit formation. Especially on the edges of parts the incorporation of lead is increased which leads to poor coverage (known as skip plating or edge pullback). Pits as well as un-plated areas lead to poor corrosion performance of the high P EN deposit. Figure 4 shows steel panels plated with THM stabilizer free high phosphorus electroless nickel at different coating thicknesses after 552h neutral salt spray test (ISO 9227) at different MTO level (Fig. 5).

Figure 5. Corrosion protection performance for THM stabilizer free high phosphorus electroless nickel on steel according to ISO 9227 NSS after 552h.
3.5 Hardness
Traditionally stabilized as well as THM stabilizer free electroless nickel deposits provide the same coating hardness. The hardness for high phosphorus electroless nickel deposits is 520-620 HV0.1, for medium phosphorus electroless nickel deposits 550-650 HV0.1.
3.6 Brightness
Cd was commonly used as brightener in electroless nickel processes especially for medium phosphorus electroless nickel deposits. The brightness provided by cadmium based electroless nickel processes is very high and can be difficult to achieve with Cd-free electroless nickel processes. Nevertheless, there is an advantage of the new THM stabilizer free medium phosphorus electroless nickel process. The deposit is fully bright and can be maintained over the complete operating bath life. The Cd containing electroless nickel processes usually decrease in deposit brightness after approx. 2 MTOs and are afterwards inferior to the THM stabilizer free medium phosphorus electroless nickel process. Therefore the new generation provides a much more consistent deposit appearance in production environment (Fig. 6).

Figure 6. Deposit appearance of THM stabilizer free medium phosphorus electroless nickel.
4. Conclusion
Environmental and legal restrictions are primary drivers in the development of new electroless nickel technology. The advanced generation of electroless nickel processes has unique formulations which are completely free of Pb, Cd and toxic heavy metal stabilizers and therefore are one step ahead of existing legislations (ELV, RoHS and WEEE).
The new THM stabilizer free electroless nickel processes are a sustainable approach, which are able to provide the same deposit performances such as uniformity of the coating, corrosion resistance and hardness as traditionally stabilized electroless nickel processes. And even in comparison to traditionally stabilized Pb and Cd containing electroless nickel processes the new electroless nickel technology offers advantages in terms of higher deposition rate, longer bath life and stable deposit brightness of medium phosphorus electroless nickel process over the complete bath life.
The results of this comparison show that the advanced electroless nickel technologies are a technically and economically reliable alternative to traditionally stabilized electroless nickel processes.
6. References
(1) Atotech technical data sheet ELeVEN HP 910
(2) Atotech technical data sheet ELeVEN MP 600
(3) Atotech standard operating procedure “BTT EN Internal Stress Test”, May 2013
(4) Atotech standard operating procedure “BTT EN Nitric Acid Test”, July 2012
(5) Atotech technical background information “BTT EN Pitting in EN deposits”, March 2013
For more information visit Atotech North America.
RELATED CONTENT
-
Qualitative Approach to Pulse Plating
In 1986, the AESF published Theory and Practice of Pulse Plating, edited by Jean Claude Puippe and Frank Leaman, the world’s first textbook on pulse plating. A compendium of chapters written by experts in this then-emerging field, the book quickly became the authoritative text in pulse plating. What follows here is the opening chapter, serving as an introduction to the field. Although the field has grown immensely in the intervening 35 years, the reader will find that the material remains a valuable introduction to those looking to advance the field of pulse plating.
-
History of Chromium Plating
A review article originally printed as Plating & Surface Finishing, 71 (6), 84-91 (1984) on the occasion of the 75th anniversary of the founding of the American Electroplaters Society.
-
Cyanide Destruction: A New Look at an Age-Old Problem
Cyanide in mining and industrial wastewaters has been around from the beginning, including electroplating processes. This presentation reviews a number of current processes, and in particular, offers new technologies for improvement in cyanide destruction by the most common process, using sodium hypochlorite.


















