Summary Report on USCAR Field Test for Trivalent Chromium
Mark Schario from Columbia Chemical explains USCAR’s multiyear project to determine how various trivalent chromium systems would perform in the real world.
#asktheexpert
Q: I understand that the United States Council for Automotive Research (USCAR) released the findings of its “Final Assessment of Decorative Trivalent Chromium Exposure in Winter Environments” study. What was decided?
A: It was a multiyear project launched to determine how various trivalent chromium systems would perform in the real world and how accelerated corrosion testing correlates with real-world results. With the goal of identifying whether chloride or sulfate-based trivalent chrome processes perform better in the areas of general and high-chloride corrosion and overall color stability, the two-phase study revealed sulfate-based systems suffered greater chrome loss in the Russian Mud corrosion performance tests. In the appearance performance tests, the study revealed the initial color advantage of the sulfate systems diminished significantly with field exposure compared to the chloride systems.
Featured Content
When looking at the overall analytical results and summary findings, the chloride-based system performed better in color stability and ranked more consistent in Russian Mud performance in the multiyear field test. Coming out of the study, it was reported that all three OEMs (FCA U.S., Ford and General Motors) recognize the advantages of chloride-based, bright trivalent chromium chemistries over sulfate.
USCAR was founded in 1992 with a goal to further strengthen the technology base of the U.S. auto industry through cooperative research and development. Its main focus is to create, support and direct U.S. cooperative research and development to advance automotive technologies, as well as be responsive to the needs of the environment and society, and include the appropriate public and private stakeholders.
The goal was also for USCAR to demonstrate the power of collaboration by enabling U.S. automakers to do great things efficiently and effectively. Individually, these same tasks would be far more difficult, potentially redundant and, in many instances, impossible to achieve as quickly by individual companies.
Under USCAR, the Decorative Trim Work Group was formed to investigate common corrosion issues affecting exterior decorative trim components. Originally formed in 2012 to discuss Russian Mud corrosion, the group includes technical specialists in the areas of materials engineering, product engineering, supplier quality and decorative finishes from Ford, General Motors and FCA U.S. As the optimum and most commercially viable alternative technology recognized for hexavalent chromium electroplating is trivalent chromium, the work group’s primary focus for 2017-19 is trivalent chrome and REACH regulations.
As reported by the work group, the field test goals included:
- Differentiation between chloride versus sulfate-trivalent chemistries.
- Performance in conventional and high chloride regions.
- Determine passivation technologies’ performance for chrome preservation and color.
- Identify the optimal trivalent chrome process (for example, chloride or sulfate) for industrywide use with respect to general corrosion, high chloride corrosion and color mastering/color stability.
The field test consisted of mounting panels on trucks and placing them in the field in the general corrosion region of Michigan and the high chloride region of Colorado. Phase I tested 17 trivalent chemistries from November to April for three consecutive years (2015-2018). Phase II tested five trivalent chemistries from November to April for two consecutive years (2016-2018).
The year three corrosion data did not provide conclusive results, so it was determined not to focus on this portion of the data. A significant and unexpected finding of the study related to the color analysis of the sulfate versus chloride trivalent systems. Color results and conclusions through year three show chloride systems were color stable on L values, while sulfate systems degraded in L values.
Overall observations were that higher chromium loss was shown on the sulfate systems. The Russian Mud test also showed higher chromium loss. USCAR also evaluated the L, a, b values of the chromium deposits before and after the field tests with appearance observations recorded in Chart 1.
Appearance Observation, Year 2. Information from the United States Council for Automotive Research.
Test results revealed that sulfate systems color degrade over time. As a result, the group felt replacement parts may not match. Chart 2 shows the color stability comparison of the three different deposits. The chloride trivalent system (shown in blue) had the best color stability over time. The hexavalent chromium (shown in black) was second for color stability. The sulfate system (shown in pink) had the poorest color stability.
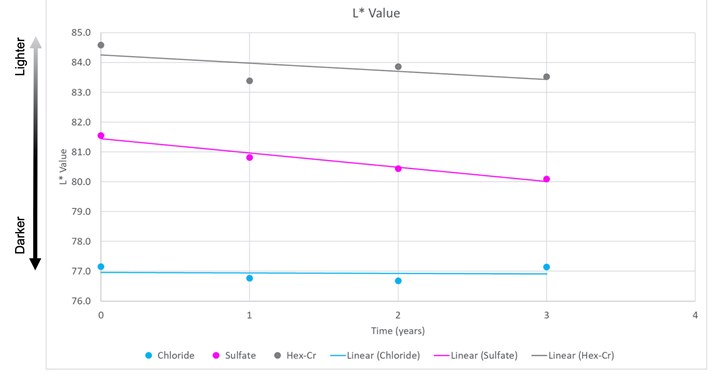
L* Value, Year 3, Pared Data. Information from the United States Council for Automotive Research.
With respect to the study’s effect of chrome loss on color data, chromium corrosion was visible on plaques from both Colorado and Michigan. High chloride chromium corrosion (Russian Mud) was more prevalent on sulfate-based chemistries than on chloride-based chemistries. The study revealed that 13 of 14 chemistries with significant chrome loss were sulfate systems.
The overall field study conclusions regarding appearance performance of sulfate versus chloride were that the color advantage of sulfate systems as plated is significantly diminished through field exposure and that color harmony of replacement parts may be a significant advantage for chloride systems.
This USCAR field corrosion study focused on performance attributes comparing the two systems to hex chrome. Because it is recognized there are other key aspects to identification of an optimum system in addition to performance, the USCAR team also considered process criteria. The three systems were evaluated by the USCAR team using a numerical evaluation matrix with 40% weight given to the process complexity and 60% given to performance as shown in Chart 3. Process and nickel leach input was received from multiple chemical manufacturers and applicators. Corrosion and appearance performance data included was based on the 3-year field study.
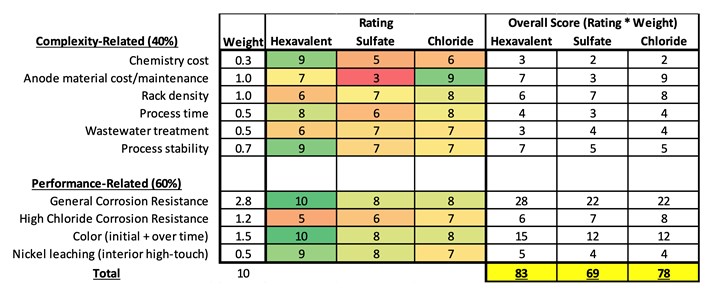
Additional Process Considerations. Information from the United States Council for Automotive Research.
FCA U.S., Ford and General Motors are committed to following the continuing worldwide regulation changes concerning hexavalent chrome and are prepared to adapt on a technical basis, as needed. Currently, designs for vehicle sales in NAFTA exclusively utilize the characteristic appearance of hexavalent chrome deposits; however, they recognize the potential need for a sudden switch. All three OEMs have published standards for allowing bright trivalent chromium; however, it must be noted that the use of decorative bright trivalent chrome chemistry must be specifically called out for any sourced component. Based on the results of this comprehensive three-year field study, all three OEMs recognize the advantages of chloride-based bright trivalent chromium chemistries over sulfate.
The information below was not part of the USCAR Field Test Study or presentation and is not to be intended as such. It is provided with the intent to offer a general overview of the usage and application of trivalent chromium and hexavalent chromium as well as a background on regulatory oversight, legislation and concerns.
Trivalent chromium has long been proven in the field. For exterior applications, trivalent has been used for many years, with the trucking industry having specified trivalent chrome plating for the past 30 years. For interior applications, trivalent has been used successfully on parts for more than 10 years. Trivalent chromium does not burn or whitewash, which significantly lowers the amount of rejects from applicators.
Hexavalent chrome plating is considered the traditional form of chrome plating, having been in use for more than 90 years. Hexavalent chromium is a known carcinogen and, as such, it is heavily regulated for both worker exposure and environmental safety. The Registration, Evaluation, Authorization and Restriction of Chemicals (REACH) regulation went into force in June 2007, regulating the usage of hexavalent chromium in the European Union with a deadline to eliminate its usage there all together. Many global manufacturers have already specified or are in the process of specifying trivalent chromium on their blueprints to meet these changing regulations.
The United States Environmental Protection Agency (EPA) continues to adopt legislation on wastewater and emissions and increase regulatory oversight of hexavalent chromium. Occupational Safety and Health Administration (OSHA)-regulated control measures have also been put in place to limit exposure to employees, which is driving up the cost of equipment and employee monitoring. Additionally, it was recognized that the compounds used in the PFOS mist suppressants needed for hexavalent plating to reduce employee exposure do not break down in nature, so the use of PFOS as a fume suppressant was banned (per request) in the surface finishing industry in 2015. Plating facilities must supply data to the EPA that they are not over discharge limits. In most cases, this involves adding extra waste treatment steps to fall within the established guidelines. The extra steps and equipment add significant costs. Finally, there is currently increasing concern, oversight and proposed legislation on the group of Per- and polyfluoroalkyl substances (PFAS) that will need close monitoring as it relates to hexavalent chromium plating. Visit NASF.org/PFAS for more resources and information on this topic.
Mark Schario is executive vice president at Columbia Chemical. Visit columbiachemical.com.
RELATED CONTENT
-
Smut and Desmutting
Question: I am new to this industry and have heard about smut and desmutting operations.
-
Choosing and Troubleshooting Copper Electroplating Processes
Learn more on this inexpensive and highly efficient process.
-
Aluminum Anodizing
Types of anodizing, processes, equipment selection and tank construction.


















