Technology Advancements for Select Key Hard Chromium Applications
This paper presents recent industry specific hard chromium technology advancements such as meeting the needs of the automotive and hydraulic industries. Examples for additional improvement potential are shown to simplify the operation and make hard chromium plating even more reliable in its specific needs for a wide range of industries served.
#research #surfin #automotive
Technology Advancements for Select
Key Hard Chromium Applications
Featured Content
A Synopsis* of a Presentation given at SUR/FIN 2018 (Cleveland, Ohio)
by
Ron Cucuro,**
MacDermid Enthone Industrial Solutions
Waterbury, Connecticut, USA
Editor’s Note: The following is a synopsis of a presentation given at NASF SUR/FIN 2018, in Cleveland, Ohio on June 4, 2018 in Session 2, Advances in Surface Finishing II. A pdf of this brief can be accessed and printed HERE; the complete Powerpoint presentation is available by clicking HERE.
ABSTRACT
It seemed like the maximum technically feasible cathodic efficiency for hard chromium processes had been achieved several years ago. Continued progress in technology is highly desired by both platers and industry to stay competitive. Given this, a more application specific approach seems the best way forward. This paper will present recent industry specific hard chromium technology advancements such as meeting the needs of the automotive and hydraulic industries. Examples for additional improvement potential are shown to simplify the operation and make hard chromium plating even more reliable in its specific needs for a wide range of industries served.
Introduction
The market for hard chromium plating is substantial. The key market segments involve (1) automotive shock absorbers, at captive facilities within OEMs; (2) hydraulics, at both job shop and captive facilities and (3) rotors and engineering applications, largely in the Americas and Europe at job and captive shops.
Electroplating of hard chromium from hexavalent chromium chemistry has been the mainstay technology for decades. Concerns have grown, however, regarding health and environmental problems with its use, and efforts toward developing alternatives have proceeded apace. While viable alternatives to decorative coatings have emerged, thicker hard chromium deposits have been more difficult to address. It is difficult to find a replacement that provides all of the desired performance properties obtained with hexavalent chromium chemistries.
On a global basis, the driver in all of this is the regulatory regimen involved with the continued use of hexavalent hard chromium. The regulations issued by REACh and OSHA dictate risk reduction to a minimal level. As seen in Fig. 1, over time, reduced exposure thresholds and emission limits have spawned various technologies for risk minimization, including the use of surfactants, enhanced ventilation and dust-free chemistries. This presentation discusses further technical risk minimization measures via the use of closed system concepts. These include (1) zero-discharge horizontal continuous plating for 6-meter hydraulic bars and (2) zero-discharge, reactor cell equipment for shock absorber plating.
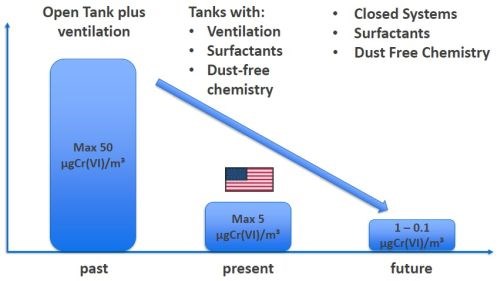
Figure 1 - REACh and OSHA rules dictate risk reduction.
Horizontal continuous plating lines for hydraulics
There are a number of issues in hard chromium plating of hydraulic rods with conventional open tanks. As shown in Fig. 2, the plating current distribution and thus the thickness distribution is poor, leading to difficulties in mechanical post-treatment (sizing, polishing, etc.). Further, a low microcrack count density produced in this cell arrangement results in poor corrosion resistance, and nodules and micropores in the deposit pose additional problems in meeting the specified 96-250 hr of salt spray (DIN EN ISO 9227).
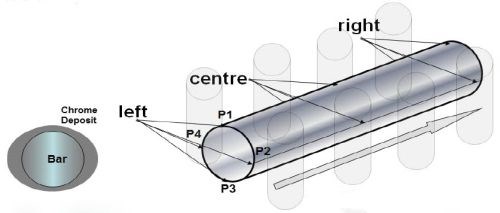
Figure 2 - Plate distribution of hard chromium on rods plated in conventional tanks.
System designs
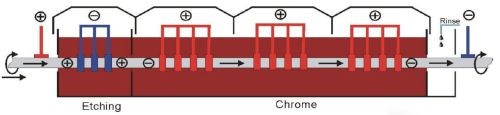
Figure 3 - Cross-section schematic of a horizontal continuous plating installation for hydraulic rods.
Horizontal continuous plating addresses the weaknesses associated with the traditional approach. As shown in Fig. 3, bars are run horizontally through the plating chamber and conforming anodes. The chamber is isolated from the outside, eliminating emissions and exposure. In addition, the rods are turned as they are passed through the cell, promoting uniform plate distribution. In this installation, the single plating tank contains 3000 L (790 gal.) of plating solution. The rods are run at 50 A/dm2 on four tracks at a line speed of 6 m/hr, or a full capacity of 24 m/hr. Figure 4 shows various line details of the horizontal hydraulic rod plating installation.

(a) Entry.

(b) Etching station.

(c) Conforming anodes (exterior view).

(d) Conforming anodes (internal view).
Figure 4 - Line details of the horizontal hydraulic rod plating installation.
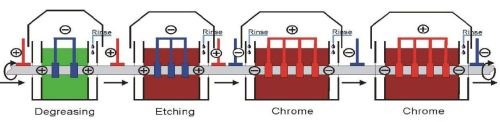
Figure 5 - Cross-section schematic of a modular horizontal hard chromium plating system.
A second modular design is shown in Fig. 5. Here the tank volume is about 14,000 L (3700 gal) for five to six plating tanks. With four to nine tracks and a line speed of 10-14 m/hr, the installation has a production capacity of 40-126 m/hr. Current densities are in the range of 50-70 A/dm2.
Bath chemistry
The substitution of platinized titanium anodes for lead has led to the development of organic catalyst systems for high efficiency hard chromium plating. As shown in Fig. 6, newly developed proprietary chemistry for hydraulic rod plating has enhanced the distribution, or throw, between conforming anodes, and has increased the plating rate by 20-30%.
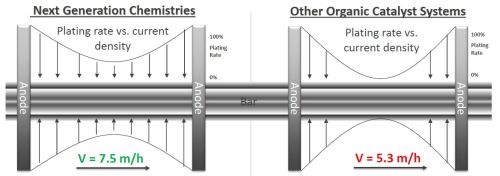
Figure 6 - Improvement in plating rate with new proprietary chemistry.

Figure 7 - Overview of continuous horizontal plating system.
Horizontal continuous hard chromium plating of hydraulic rods (Fig. 7) offers a closed system with zero emissions and worker exposure, in compliance with future regulations. The system has the ability to plate to size, eliminating dog-boning effects of poor plate distribution. The bath chemistry is 20-30% faster than conventional solutions, allowing higher productivity. There is logistical efficiency with the system; the plant can be fully integrated between pre- and post-grinding. Finally, the concept is fully commercial and is becoming more dominant in the 6-meter bar market.
Improved automation for shock absorber plating
Hard chromium plating for shocks and struts has evolved over the years (Fig. 8). Before 1950, conventional rack lines were used, with open tanks and return-type machines. Although the occupational health problems would not be addressed until later, production suffered from poor thickness distribution, which required expensive post-grinding operations, adding to already-high plating costs.

Figure 8 - Evolution of hard chromium plating technology for shock absorbers and struts.
By the end of the 20th century, the health issues were being addressed and the operational standard used enclosed rack lines with platinized titanium anodes. Although the conditions outside the enclosure were clean and safe, the plating equipment inside the enclosure was contaminated with hexavalent chromium aerosols from the open tanks. Still, plating costs were reduced.
Today, emission-free technology is available to address the regulatory challenges presented by REACh. The modular system if Fig. 8, utilizes a reactor cell under vacuum. The result is zero discharge, with no air or water contamination.
In the emission-free system, there is a single rectifier dedicated to each part, eliminating part-to-part deviation. The degree of emission and exposure elimination promises compliance with any future regulations foreseen today. The precision of thickness control and logistical efficiency yields further reduction in plating costs. Finally, the resulting coatings have been approved by major automotive OEMs for neutral salt spray performance, microcracks and thickness specifications.
Summary
In the global key market segments, there will be continued future demand for low cost, high performance, wear and corrosion-resistant coatings, such as hard chromium. With growth in zero discharge technology, the risk of using hexavalent chromium can be reduced to zero. This will allow the applicators to meet even tighter regulations, while staying competitive and improving their bottom lines. The benefits of these hard chromium coatings outweigh the risks associated with hexavalent chemistry.
About the author / presenter
Ron Cucuro is Hard Chrome & Surface Finishing Product Manager North America for MacDermid Enthone Industrial Solutions, Waterbury, Connecticut.
*Compiled by Dr. James H. Lindsay, Technical Editor - NASF
** Corresponding author:
Ron Cucuro
Hard Chrome & Surface Finishing Product Manager North America
MacDermid Enthone Industrial Solutions
245 Freight Street
Waterbury, CT 06702
E-mail: Ron.Cucuro@macdermidenthone.com
RELATED CONTENT
-
Hybrid Sol-Gel Coatings in Surface Engineering
A look at the use of modified sol-gel polymer films and hybrid system coatings, as well as the methodologies for evaluating the mechanical properties of the coatings.
-
Methods and Formulas to Determine Internal Deposit Stress in Applied Metallic Coatings
Internal stress exists in electroplated and chemically applied metallic coatings. This paper reviews the test procedures for measuring deposit stress and the formulas used to calculate stress values. Many formulas used require modification to obtain actual internal stress values. Errors in this regard are examined and common mistakes are explained.
-
A Process for Alkaline Non-cyanide Silver Plating for Direct Plating on Copper, Copper Alloys and Nickel Without a Silver Strike Bath
Traditionally, silver is electroplated in toxic, cyanide-based chemistry. Due to cyanide’s extreme hazard to human health and environments, developing non-cyanide silver chemistry is essential for the silver electroplating industry. Discussed here is an aqueous, alkaline non-cyanide silver plating technology, which can be directly plated over nickel as well as copper and its alloys. The silver deposits have perfect white color and better anti-tarnishing properties than other non-cyanide silver processes. The silver is plated entirely from the dissolving silver anode and the bath is very stable, and maintains a stable pH level both during plating and idle time. This new non-cyanide silver technology will plate bright silver that is perfectly suitable for electronic, industrial and decorative applications. .


















