Electrodeposited Inconel and Stellite-like Coatings for Improved Corrosion Resistance in Biocombustors
This work deals with problems in the Third World – namely improving the safety and efficiency of cookstoves used by people around the world. A scalable and economic process is required to apply coatings on low-cost stainless steel substrates for enhanced high temperature corrosion resistance specifically targeted towards biomass combustion apparatus. Cost effective, scalable, and flexible electrodeposition-based coating of various alloys [Ni/Co]-Cr-[Mo/Fe] system, that are able to withstand high temperature corrosion and improve the functional lifetime of existing and next generation bio-combustors components is a desirable technological advancement. Within this context, a wide array of electrolytes and processing parameters were evaluated to develop an ideal alloy coating.
#research #surfin
A Synopsis* of a Presentation given at SUR/FIN 2017 (Atlanta, Georgia)
by
Featured Content
S. H. Vijapur (Presenter),** T. D. Hall, E. J. Taylor, M. E. Inman
Faraday Technology, Inc.
Englewood, Ohio, USA
and
M. Brady (ORNL)
Editor’s Note: The following is an updated synopsis of a presentation given at NASF SUR/FIN 2017, in Atlanta, Georgia on June 19, 2017 in Session 2, Advances in Surface Finishing Technology I. This brief can be accessed and printed HERE; the complete Powerpoint presentation is available by clicking HERE.
ABSTRACT
A scalable and economic process is required to apply coatings on low-cost stainless steel substrates for enhanced high temperature corrosion resistance specifically targeted towards biomass combustion apparatus. Cost effective, scalable, and flexible electrodeposition-based coating of various alloys [Ni/Co]-Cr-[Mo/Fe] system, that are able to withstand high temperature corrosion and improve the functional lifetime of existing and next generation bio-combustors components is a desirable technological advancement. Within this context, a wide array of electrolytes and processing parameters were evaluated to develop an ideal alloy coating. Specifically, NiCr and NiCoCr binary alloy coatings demonstrated enhanced corrosion resistance when exposed to an aggressive environment (~700°C, 1000 hr, coating surface salted with ~3 mg/cm2 every 100 hours) on different SS substrates. When compared to the SS base material the NiCr and NiCoCr coating exhibited a 70% weight loss and 3.4 times lifetime improvement over its base material.
Introduction
This particular work is laudable in that it deals with problems in the Third World – namely improving the safety and efficiency of cookstoves used by people around the world. According to the Global Alliance for Clean Cookstoves:
“The use of open fires and traditional cookstoves and fuels is one of the world's most pressing health and environmental problems. Globally, three billion people rely on solid fuels to cook, causing serious environmental and health impacts that disproportionally affect women and children. According to the World Health Organization, household air pollution from cooking kills over 4 million people every year and sickens millions more” (Fig. 1).

Figure 1 – Typical cookstoves.
With this work, Dr. Vijapur has worked to create more efficient, longer lasting, cleaner and cost effective cookstoves for use in burning biomaterials. Biomass combustion reactors are subjected to various corrosive attacks due to the wide range of potential materials that will be burned in them, including gasoline, grass and other carbon-based waste, which give off prodigious amounts of CO, CO2 and other carcinogens. To meet these problems, it is necessary to increase burning efficiency, burn at high temperatures and design low cost material systems to durably perform in these environments. To address the latter problem, Dr. Vijapur’s approach was to apply a high value coating to existing bio-combustors or lower cost steels.
The objective was to develop a cost effective, scalable, and flexible electrodeposition-based coating process that could be applied to the corrosion sensitive regions of existing and next generation bio-combustors. The coating could consist of corrosion-resistant alloys such as Stellite 21 (Co-base, Ni-3%, Cr-29%, Mo-6%) or Inconel 740 (Ni-base, Co-20%, Cr-25%, Mo-0.5%). Considering possible alloy coatings:
- Nickel- and cobalt-based alloys have been shown to possess excellent alkali/halide attack resistance.
- Cobalt-based alloys have been shown to possess sulfidation resistance.
- Molybdenum additions into the metal alloys also seem to improve corrosion resistance and high temperature stability.
- Chromium improves temperature stability and corrosion resistance in high temperature systems.
Electrolyte development
Keeping in mind that costs were a major consideration in of any modification of a bio-combustor, low cost ferritic or austenitic steels were used as test substrates. A Hull cell was used to determine the solution composition and processing conditions which would deposit binary and ternary alloys of the family [Ni-Co]-Cr-[Mo-Fe]. A standard trivalent chromium electrolyte was used with varying amounts of the other metals. X-ray fluorescence was used to determine the deposit composition through the current density range along the Hull cell panel.
Commonly used in production control, the venerable Hull cell remains an invaluable tool in electrodeposition science and engineering. In this case compositional screening of Ni-Cr and Ni-Co-Cr alloys showed that a wide range of potential deposit composition could be obtained by controlling the nickel concentration (Fig. 2). Similar results are shown for Ni-Cr-Fe ternary deposits, both with DC and pulse waveforms (Fig. 3).

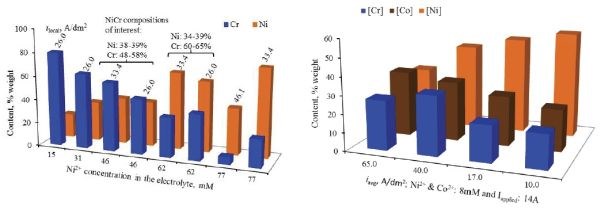
Figure 2 – Ni-Cr (L) and Ni-Co-Cr (R) alloy content versus nickel concentration in solution at various local current densities on the Hull cell specimens.
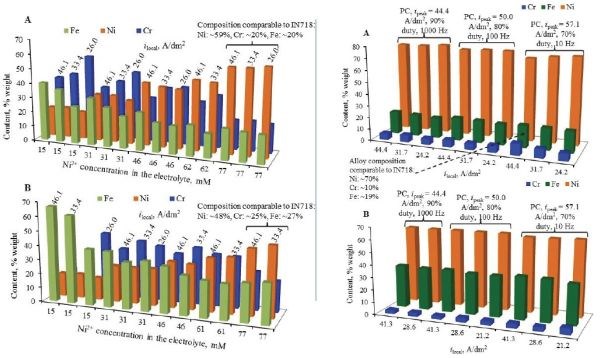

Figure 3 – (L) Ni-Cr-Fe alloy content versus nickel concentration in solution at various local current densities on the Hull cell specimens; (R) results with pulse waveforms.
Adding molybdenum, it was found that alloys approaching the compositions of Inconel 740 and Stellite® 21 could be produced (Fig. 4).
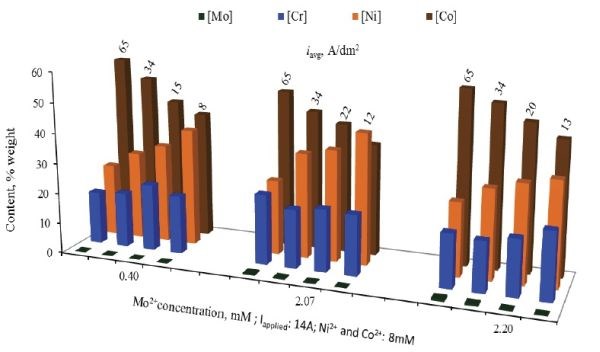

Figure 4 – Results of compositional Hull cell screening of quaternary Ni-Co-Cr-Mo alloys.
Coupon studies
Using the electrolyte designed within the Hull Cell study and approximate applicable current densities we transitioned to coupon studies. 1” × 4” samples were plated with different deposits in a Faradayaic® flow chamber, providing uniform laminar solution flow. Pure nickel, pure chromium, Ni-Cr, Ni-Cr-Fe, Ni-Co-Cr and Co-Cr deposits were studied at varying thicknesses and, in the case of nickel, varying nickel concentrations in solution. The results are shown in Fig. 5.


Figure 5 – Coupon samples as-plated.
Corrosion studies
Corrosion studies of the alloy deposits consisted of 100 hr of salted oxidation with high salt loading (~3 mg/cm2). A 3.5 wt% solution of sea salt was applied to the surfaces before corrosion testing. Ten 100-hour cycles of salted oxidation, involving an initial thermal cycling regimen was used. The results for the high-level salt exposure are shown in Fig. 6.
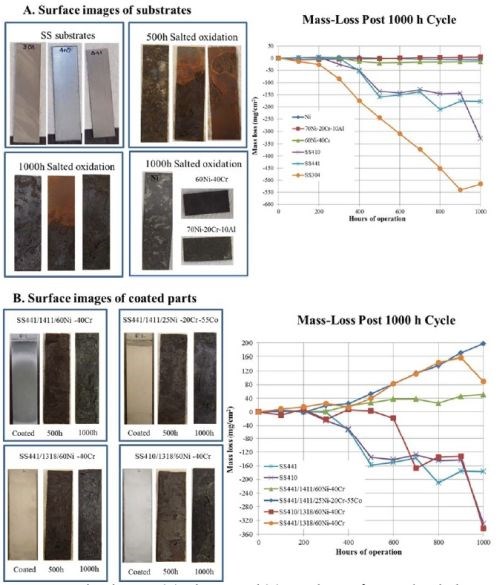
Figure 6 - Comparison between (A) substrates and (B) coated parts after 1000 hr salted oxidation trials [3 mg/cm2].
The results of the entire study indicated that the pure chromium and nickel deposits did not maintain adhesion during the corrosion studies. However, the ~60/40 Ni-Cr and Ni25-Co55-Cr20 alloy deposits exhibited the best corrosion resistance, albeit dependent on thickness. The ternary (Ni/Cr/Fe) deposits did not maintain adhesion during the oxidation study, likely due to a mismatch of coefficients of thermal expansion (CTE).
As for appropriates to their use in bio-combustors, the ~50 wt% Ni-Cr alloys improved the corrosion resistance of the base stainless-steel substrate by at least 70%, potentially leading to a 3.4-fold improvement in cook stove lifetime. An initial economic analysis indicated that a 4-mil coating unit cost declined with the number of units produced: (a) 10,000 units - $15.03; (b) 100,000 units - $2.91; (c) 1,000,000 units - $1.69.
Summary
Summing it up, this work:
- Demonstrated a wide range of [Co/Ni]–Cr–[Mo/Fe] alloy coating compositions that can be produced
- Demonstrated process scalability from a Hull Cell to flat coupons.
- Demonstrated the potential of specific coatings to surpass 1000 hours of accelerated high temperature corrosion testing
- Showed that 50 wt% NiCr alloys improve corrosion resistance of the base stainless-steel substrate by at least 70%, potentially leading to a 3.4 fold cookstove lifetime improvement.
- Demonstrated that the process may be an economically viable approach for improving cookstove bio-combustor lifetimes.
- Showed a cost estimate that for 50 wt% NiCr coating to be $2.93/in.3 or $1.69 for a 4 mil coating on a 0.61 ft2 component produced at 1,000,000 units per year, realizing an 89% cost reduction compared to base IN625 materials.
About the author / presenter


*Compiled by Dr. James H. Lindsay, NASF Technical Editor, and updated by Dr. Vijapur.
** Presentation author:
Santosh H. Vijapur, Principal Scientist
Faraday Technology Inc.
315 Huls Dr.
Englewood, OH 45315
PHONE: (937) 836 7749
EMAIL: santoshvijapur@faradaytechnology.com
RELATED CONTENT
-
Cyanide Destruction: A New Look at an Age-Old Problem
Cyanide in mining and industrial wastewaters has been around from the beginning, including electroplating processes. This presentation reviews a number of current processes, and in particular, offers new technologies for improvement in cyanide destruction by the most common process, using sodium hypochlorite.
-
Black Chromium Finishing: Beauty and the Beast
Over the past 10 years, there has been commercial development of black chromium deposits from a trivalent chromium electrolyte. This paper will review the deposit characteristics and operational consideration of these similar, but different chromium plating deposits.
-
Chromium-free Etching and Palladium-free Plating of Plastics
Plastics are replacing metals in the manufacture of many parts, and quite often there is a need for metallic coatings on the plastics and other non-conductors. This paper will describe new processes of preparing ABS plastic substrates for subsequent metallization.


















